Acutetraumatic spinal cord injury (SCI) is linked to an elevated risk of venous thromboembolic complications, including deep vein thrombosis (DVT) and pulmonary embolism (PE).1The incidence of venous thromboembolic events among patients with acute SCI who do not receive any prophylaxis or receive suboptimal prophylaxis ranges from 4% to 100% after accounting for variations in injury level, diagnostic methods, and surveillance strategies.2Decision-making concerning thromboprophylaxis for these patients is often multifactorial, as factors such as immobility, the existence of polytrauma, and the need for surgical interventions contribute to their complexity.
The literature extensively describes the efficacy of venous thromboembolism (VTE) chemoprophylaxis in trauma.3–5However, the risk of hemorrhagic complications following anticoagulation administration in patients with trauma can be a significant concern, particularly in those with spine fractures and scheduled operative procedures for their injuries due to the risk of postoperative epidural hematoma. Consequently, patients with trauma frequently experience delays in chemoprophylaxis administration or interruptions in doses.6Studies indicate that initiating prophylaxis early and continuing it for approximately 3 months postinjury are effective strategies for preventing VTE.7–9Several published investigations have reported the outcomes of individual or combined prophylactic measures.10–14这些方案有很大的波动,使其challenging to establish general recommendations. Nonetheless, the collective findings suggest that some prophylaxis is superior to no prophylaxis and that certain pharmacological prophylaxis options may be more effective than none at all.
Evidence shows that initiating chemoprophylaxis closer to the time of injury is associated with lower rates of DVT and VTE,15–18and therefore several systematic reviews recommend starting chemoprophylaxis within 72 hours of SCI.18–21Two guidelines recommend initiating prophylaxis within the first 72 hours after SCI.19,20,22,23Low-molecular-weight heparin (LMWH) is the recommended chemoprophylaxis agent in both guideline studies.19,20,22,23Although several studies have compared the efficacy of different chemoprophylaxis agents,15,17few studies have examined the timing of prophylaxis and its relationship to clinical outcomes. The objective of this study was to investigate the safety profile of a protocol for initiating VTE chemoprophylaxis within 24 hours of injury or surgery and to compare prophylactic results with those reported previously. We determined in-hospital rates of VTE and hemorrhagic complications in a cohort of patients with acute SCI and evaluated the variables that predict VTE in this cohort.
开云体育世界杯赔率
Study Population and Criteria
Beginning in 2015, patients were enrolled into the Transforming Research and Clinical Knowledge in SCI (TRACK-SCI) study, a multi-institutional prospective observational study as previously described.24This study was approved by the Committee on Human Research of the University of California, San Francisco. Patients in this study provided consent for treatment and for publication. Patients were enrolled after presenting to the emergency department with a traumatic SCI. Demographic, clinical, biospecimen, imaging and outcome data were collected and stored in a REDCap database.25Inclusion criteria for this study included patients with traumatic SCI who were treated under institutional SCI guidelines, admitted to the intensive care unit (ICU), and 18 years of age or older at the time of injury. Surgical and medical treatment were directed by the discretion of the attending physicians. We excluded patients with American Spinal Injury Association (ASIA) grade E upon admission. The standard protocol was administration of chemical DVT prophylaxis (40 mg of subcutaneous enoxaparin daily) within 24 hours of injury, or within 24 hours of SCI surgery for patients who underwent operative intervention for SCI. For patients who needed revision surgery, follow-up surgery, or had a bleeding event, LMWH was delayed for at least 24 hours, with the length of LMWH interruption based on the treating clinician’s clinical judgment.
Outcome Variables
Primary outcomes of interest included in-hospital rates of VTE (including DVT and PE) and in-hospital hemorrhagic complications that occurred after LMWH administration. Although patients did not undergo routine VTE screening, duplex ultrasound was utilized if a DVT and/or PE was clinically suspected. CT angiography was used to evaluate for PE. In-hospital hemorrhagic complications were defined as any instances of worsening of bleeding or new bleeding during hospitalization occurring after LMWH administration. In-hospital hemorrhagic complications were categorized as: postoperative SCI surgery bleeding, lumbar drain (LD) bleeding complications, bleeding due to tracheostomy tube, bleeding due to percutaneous endoscopic gastrostomy (PEG) tube or nasogastric (NG) tube, worsening of intracranial hemorrhage (ICH; defined as interval worsening or increase on imaging), postoperative non-SCI surgical bleeding, other nonsurgical bleeding, and wound dehiscence. Secondary outcomes included ICU and hospital length of stay (LOS), discharge location type, and in-hospital mortality.
Statistical Analysis
Descriptive statistics were used to characterize the demographics, clinical characteristics, and outcomes of our cohort. Patients without data for a variable of interest were excluded from the calculation of percentages for that particular variable. Univariate regression analysis was utilized to identify variables associated with VTE. A p value < 0.05 was used as the threshold of statistical significance. Statistical tests were performed using Python and the following libraries: pandas (version 1.5.3), NumPy (version 1.20.3), and statsmodels (version 0.13.5). To conduct a literature search on past studies examining the effects of timing of VTE prophylaxis on VTE rates and bleeding rates, we searched in PubMed using the following keywords: "low molecular weight heparin", "spinal cord injury", and "timing" (Table 1).
Summary of previous studies examining the effects of chemoprophylaxis on VTE and bleeding complication rates in SCI patients
| Authors & Year | Hrs from SCI/Surgery That CP was Administered | Study Design | VTE Rates | Bleeding Complications | Routine or Symptomatic Monitoring of VTE |
|---|---|---|---|---|---|
| Aito et al., 200226 | W/in 72 hrs of injury | Direct comparison of VTE rates btwn 99 acute SCI pts given LMWH w/in 72 hrs vs those in 176 SCI pts w/ delayed LMWH admin 8–28 days after injury | DVT 2% in those receiving LMWH w/in 72 hrs of injury was significantly < than the DVT rate in those w/ delayed LMWH admin (26%) | Not examined | Routine monitoring (on admissions & after 45–60 days) |
| Spinal Cord Injury Thromboprophylaxis Investigators, 200314 | W/in 72 hrs of SCI | 230 SCI pts randomly assigned to receive unfractionated heparin & enoxaparin, 246 received unfractionated heparin & intermittent pneumatic compression; VTE & bleeding rates compared btwn the 2 groups | No differences in VTE (63.3% vs 65.5%) while pts who received enoxaparin had lower PE rates (5.2% vs 18.4%) | No differences in bleeding rates (5.3% vs 2.6%) | Routine monitoring (blood work on day 2, 7, & 14; duplex ultrasonography on day 14 or w/in 2 days of last medication dose during acute phase) |
| Green et al., 199017 | W/in 72 hrs of SCI (given ≥24 hrs after injury; if surgery required, VTE prophylaxis administered the following day) | 21 SCI pts administered standard heparin & 20 administered LMWH; VTE & bleeding rates compared btwn the 2 groups | VTE 23.8% in standard heparin group, 0% in LMWH group | Bleeding complication rate 9.5% in standard heparin group, 0% in LMWH group | Routine monitoring (VTE symptoms, scheduled labs & imaging) |
| Green et al., 199427 | W/in 72 hrs of SCI | LMWH administered to 48 pts, VTE & bleeding complication rates examined | During trial, DVT 12.5% & PE 2.1% | Bleeding complication rate 2.1% | Routine monitoring (regular schedule for venous ultrasonography) |
| Merli et al., 199228 | W/in 48 hrs of SCI | Comparison of VTE btwn 19 SCI pts receiving low-dose heparin vs 17 control SCI pts | Rate of DVT in those given low-dose heparin w/in 28 hrs of SCI = 5.3%, & 35.3% in control group | Not examined | Routine monitoring (125-I fibrinogen scanning daily for 2 wks) |
| Chang et al., 201715 | W/in 48 hrs of injury | Comparison of VTE & expanding intraspinal hemorrhage rates btwn 260 SCI pts w/ early admin of heparinoids and/or aspirin (w/in 48 hrs of injury) vs those in 241 SCI pts w/ no earlier initiation of VTE prophylaxis | VTE 5% in pts receiving early VTE prophylaxis, 9% in those who did not; early admin of heparinoids associated w/ reduced VTE rates, but early admin of aspirin was not | Neither heparinoid nor aspirin admin associated w/ expanding intraspinal hemorrhage | Diagnostic imaging based on symptoms |
| Zeeshan et al., 201829 | W/in 48 hrs after surgery | 静脉血栓栓塞、出血并发症发生率的比较1772 SCI pts given anticoagulants (either heparin or LMWH) w/in 48 hrs after surgery vs those in propensity-matched 1772 pts given anticoagulants after 48 hrs from surgery | DVT 2.1% in SCI receiving anticoagulants w/in 48 hrs after surgery vs 10.8% in those receiving anticoagulants after 48 hrs from surgery; no differences in PE rates btwn groups | Bleeding rate*1.3% in SCI pts receiving anticoagulants w/in 48 hrs after surgery vs 1.2% in those receiving anticoagulants after 48 hrs from surgery (not statistically different); no differences btwn the 2 groups in intraspinal hematoma development rates secondary to anticoagulation (0.9% vs 1.5%) | Unknown (data collected from the ACS TQIP database) |
| Jacobs et al., 201330 | Average of 2.4 days postop (range 1–14 days) | 静脉血栓栓塞、出血并发症发生率的比较SCI 171 pts receiving CP w/in 48 hrs of surgery vs rates in 56 pts who did not receive any CP | 静脉血栓栓塞发生率高的那些收到CP w /48 hrs of surgery vs those who received no CP (7% vs 14.3%) | No epidural hematomas in both groups; 1.8% of pts given CP underwent irrigation & debridement for wound drainage vs 5.3% of pts w/ no CP | Monitoring of VTE based on clinical suspicion |
| DiGiorgio et al ., 201731 | W/in 24 hrs of SCI | VTE & bleeding complication rates in 49 SCI pts given LMWH w/in 24 hrs of SCI | DVT 6.1%, PE 4.1%, & VTE 10.2% | No bleeding complications | Monitoring of VTE based on clinical suspicion |
| Ahlquist et al., 202032 | W/in 24 hrs of surgery vs 24–72 hrs after surgery | VTE & bleeding complication rates in 49 SCI pts receiving either LMWH or unfractionated heparin w/in 24 hrs of surgery vs 20 pts receiving VTE prophylaxis w/in 24–72 hrs after surgery | VTE different btwn the 2 groups (6.1% vs 35%) & DVT also different (4.1% vs 30.0%); no statistical differences in PE btwn groups (6.1% vs 5.0%) | No bleeding complications | Monitoring of VTE based on clinical suspicion |
ACS = American College of Surgeons; admin = administration; CP = chemoprophylaxis; pts = patients; TQIP = Trauma Quality Improvement Program.
Defined as the need for prophylaxis blood transfusion.
Results
Patient Demographics
One hundred sixty-two patients were included in this study. Fifteen of the 162 patients withdrew from the study, leading to loss of data for certain variables for these patients. In our patient population, 28% were female and the average age (± SD) was 56.9 ± 18.5 years (Table 2).Most patients identified as White (26.5%) or Asian (21.6%) and most were not Hispanic or Latino (83%). The average Injury Severity Score (ISS) on arrival was 24.7 ± 13.8. Most patients had a cervical SCI level (81%) and most had an ASIA grade of D on admission (51.2%). The average bilateral upper-extremity ASIA motor score on admission (± 2 days) was 30.1 ± 18.1, the average bilateral lower-extremity ASIA motor score on admission was 26.9 ± 21.6, and the average total-extremity ASIA motor score on admission was 57.4 ± 33.3. Nonneurosurgical injuries on admission, which we defined as having a fracture in the extremities, were found in 18.1% of the patients.Table 2lists details of the patient demographics and initial presentation recorded in our study cohort.
Patient demographics and initial presentation
| Variable | Value (%) |
|---|---|
| No. of patients | 162 |
| Mean age ± SD, yrs | 56.9 ± 18.5 |
| Sex | |
| Male | 108 (72) |
| Female | 42 (28) |
| Unknown | 12 |
| Race | |
| American Indian or Alaska Native | 1 (0.6) |
| Asian | 35 (21.6) |
| Black or African American | 27 (16.7) |
| Hispanic | 15 (9.3) |
| Native Hawaiian or Other Pacific Islander | 2 (1.2) |
| Other | 10 (6.2) |
| White | 43 (26.5) |
| Unknown | 29 (17.9) |
| Ethnicity | |
| Hispanic or Latino | 25 (17) |
| Not Hispanic or Latino | 122 (83) |
| Unknown | 15 |
| Mean ISS on arrival ± SD | 24.7 ± 13.8 |
| SCI level | |
| Cervical | 115 (81) |
| Thoracic | 20 (14.1) |
| Lumbar | 7 (4.9) |
| Unknown | 20 |
| ASIA grade on admission* | |
| A | 32 (24.8) |
| B | 11 (8.5) |
| C | 20 (15.5) |
| D | 66 (51.2) |
| Unknown | 33 |
| ASIA motor score on admission ± SD* | |
| Upper bilat extremity | 30.1 ± 18.1 |
| Lower bilat extremity | 26.9 ± 21.6 |
| Total bilat extremity | 57.4 ± 33.3 |
| Nonneurosurgical injuries† | |
| Yes | 27 (18.1) |
| No | 122 (81.9) |
| Unknown | 13 |
On admission ± 2 days.
Extremity fractures.
Patient Procedures
One hundred thirty patients underwent decompression and/or fusion surgery after SCI (87.8%), 5 patients went to the operating room (OR) only for non–SCI-related procedures (3.4%), and 13 patients did not go to the OR for any procedures (8.8%). Most patients did not have an LD placed during their hospital stay (77.7%), while 22.3% of patients underwent LD placement (Table 3).
Patient procedural status
| Variable | No. (%) |
|---|---|
| Surgery status | |
| Underwent decompression &/or fusion surgery for SCI | 130 (87.8) |
| Did not go to OR for any procedure | 13 (8.8) |
| Only went to OR for non-SCI procedures only | 5 (3.4) |
| Unknown surgery status | 14 |
| LD use | |
| Yes | 33 (22.3) |
| No | 115 (77.7) |
| Unknown | 14 |
Patient Outcomes
A DVT occurred in 11 (7.4%) of the 148 patients who had DVT data available, while 9 (6.1%) of the 148 patients who had PE data available had a PE. Overall, 18 (12.2%) of the 148 patients with VTE data available experienced a VTE event (Table 4).This rate is within the lower range reported previously in the SCI literature (Table 1).平均ICU和洛杉矶为9.1±9.2天average hospital LOS was 14.8 ± 13.4 days. Most patients were discharged to either acute rehabilitation (32%) or a skilled nursing facility (SNF; 30%). Eight patients (5.3%) died in the hospital (Table 5).Of the 8 patients who died in-hospital, 2 also experienced a VTE. One of the 2 patients had a PE and was on a heparin drip, subsequently developing an epidural hematoma. However, the patient ultimately died from the PE. We cannot rule out LMWH administration contributing to the hematoma, but it likely expanded with the heparin drip that was started due to the PE. The second patient also suffered from a PE, but a heparin drip was not given at the time due to a recent operation. However, the patient later died from cardiac arrest and shock that was refractory to maximal pressors.
VTE, DVT, and PE rates
| Variable | No. (%) |
|---|---|
| DVT | |
| Yes | 11 (7.4) |
| No | 137 (92.6) |
| Unknown | 14 |
| PE | |
| Yes | 9 (6.1) |
| No | 139 (93.9) |
| Unknown | 14 |
| VTE | |
| Yes | 18 (12.2) |
| No | 130 (87.8) |
| Unknown | 14 |
Other patient outcomes
| Variable | Value |
|---|---|
| Mean ICU LOS ± SD, days | 9.1 ± 9.2 |
| Mean hospital LOS ± SD, days | 14.8 ± 13.4 |
| Discharge location type, n (%) | |
| Acute rehabilitation | 48 (32) |
| Deceased | 8 (5.3) |
| Home | 23 (15.3) |
| Hospice | 1 (0.7) |
| Hotel | 1 (0.7) |
| Nursing home | 1 (0.7) |
| Respite care/shelter | 2 (1.3) |
| SNF | 45 (30) |
| Transfer to acute hospital/medical ICU | 21 (14) |
| Unknown | 12 |
| Mortality rate, n (%) | 8 (5.3) |
Predictors of VTE
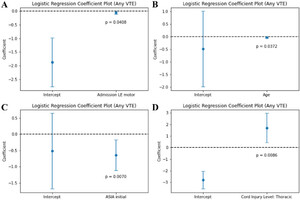
Univariate regression analysis showed that worse lower-extremity motor score at admission (p = 0.0408), worse admission ASIA grade (p = 0.0070), and younger age (p = 0.0372) were associated with development of a VTE. The "other" race category (p = 0.0052) and injury at the thoracic level (p = 0.0086) were also associated with a VTE (Fig. 1).

Graphs of univariate regression analysis between variables and VTE: admission lower-extremity (LE) motor score and VTE (A; p = 0.0408), age and VTE (B; p = 0.0372), initial ASIA grade and VTE (C; p = 0.0070), and injury at the thoracic level and VTE (D; p = 0.0086).
Complications
There were 16 overall cases of bleeding complications (10.9%) that occurred after LMWH administration in the 147 patients with data available for this variable (Table 6).Of the 127 patients who underwent SCI surgery with data available for bleeding complications, there were 3 cases of postoperative SCI bleeding (2.4%) after LMWH initiation, with 1 case (0.8%) requiring return to the OR for epidural hematoma evacuation. Another case of postoperative bleeding after SCI surgery required return to the OR, but this occurred before LMWH administration. Thirteen (8.8%) of the 147 patients had a bleeding complication not related to spine surgery. There were no bleeding complications associated with LD or tracheostomy tube placement. There were two instances of bleeding due to NG tube placement, but none due to PEG tube placement. Of the 147 patients with ICH data available, 19 (12.9%) had ICH on admission, but none had an interval worsening or increase in the size of hemorrhage that occurred after LMWH administration. Three (15.8%) of the 19 patients had interval worsening or an increase in ICH size, but these occurred before LWMH was administered. These 3 patients were followed up to ensure stability, as it is our protocol to document radiographic stability with a follow-up head CT scan within 4–8 hours.
Hemorrhagic complication rates
| Variable | Value (%) |
|---|---|
| Any bleeding complication | |
| Yes | 16 (10.9) |
| No | 131 (89.1) |
| Unknown | 15 |
| Postop SCI surgery bleeding | |
| Yes | 3 (2.4) |
| No | 124 (97.6) |
| Unknown | 3 |
| Postop SCI surgery bleeding requiring return to surgery | |
| Yes | 1 (0.8) |
| No | 126 (99.2) |
| Unknown | 3 |
| Any bleeding not related to spine surgery | |
| Yes | 13 (8.8) |
| No | 134 (91.2) |
| Unknown | 15 |
| LD bleeding complication | |
| Yes | 0 (0) |
| No | 147 (100) |
| Unknown | 15 |
| Bleeding due to tracheostomy tube | |
| Yes | 0 (0) |
| No | 147 (100) |
| Unknown | 15 |
| Bleeding due to PEG or NG tube | 2 |
| ICH present on admissions | |
| Yes | 19 (12.9) |
| No | 128 (87.1) |
| Unknown | 15 |
| Worsening of ICH | |
| Before LMWH administration | 3 |
| After LMWH administration | 0 |
| 出血后non-SCI杂志ery | 3 |
| Other nonsurgical-related bleeding | |
| Yes | 8 (5.4) |
| No | 139 (94.6) |
| Unknown | 15 |
| Wound dehiscence | |
| Yes | 0 (0) |
| No | 147 (100) |
| Unknown | 15 |
| Hardware misplacement/failure | 2 |
有3出血并发症的实例fter non–spine-related surgery. One case involved a retained hemothorax after washout, which required returning to the OR for video-assisted thoracoscopic surgery. The second case involved a hematoma on the thigh due to an intramedullary rod placement. The third case involved an enlarged hematoma near the pubic symphysis after a pelvic open reduction and interval fixation. There were 8 cases of bleeding not related to any surgery (5.4%) of the 147 patients with bleeding complication data available. Of these 8 cases, 2 involved worsening of existing or the development of a new hemothorax, 1 was an interval development of a subdural hematoma in the lumbosacral region, 1 was an interval development of an epidural hematoma in the cervical region, 2 patients developed hematuria (1 due to foley catheter placement and another due to cystine crystals in urine), and 2 patients had a gastrointestinal hemorrhage. There were 2 instances of hardware misplacement or failure, with both requiring return for revision surgery. No patients experienced wound dehiscence during hospitalization (Table 6).
Discussion
The timing of chemoprophylaxis for acute SCI is an important clinical question due to the high risk of VTE in these patients, and currently there is limited evidence to support one timing practice over another. Several prior studies have reported safety and efficacy of initiating LMWH within 72 hours of injury or surgery,14,17,26,27within 48 hours,15,28–30and within 24 hours31,32(Table 1).One study specifically found that VTE chemoprophylaxis did not increase postoperative complications in high-risk patients and that patients with VTE had a longer time to initiating chemoprophylaxis after operative fixation compared with those who did not develop VTE.33In the present study, we analyzed a cohort of 162 patients with SCI who received LMWH within 24 hours of injury or surgery and observed a 12.2% rate of VTE, which is within the lower range of previously reported rates evaluating for symptomatic VTE (9%–26%). Given the lack of evidence to support routine VTE surveillance,2the present study protocol consisted of diagnostic testing for symptomatic VTE. Our results show that initiation of chemoprophylaxis within 24 hours of injury or surgery may provide benefits for preventing VTE but may come at a cost of a low to moderate risk of increasing hemorrhagic complications in patients with acute SCI.
Within our cohort, 19 patients had ICH on admission. However, none of these patients developed interval worsening or increased size in ICH after LMWH administration. Given the risk of a worsening ICH after chemoprophylaxis, it is important to weigh the risks and benefits before initiating chemoprophylaxis. Previous studies show that early administration of chemoprophylaxis (≤ 24 hours of injury) has the same risk of worsening ICH compared with later administration (> 24 hours after injury) in patients with traumatic brain injury.34,35Our rate of ICH worsening after LMWH administration is lower than most of the previously reported ranges (0%–18%),5,34,35therefore our results suggest that early initiation of LMWH does not significantly increase the risk of worsening ICH in polytrauma patients, given that their ICH is found to be stable on follow-up brain CT. However, because our study found a low to moderate increased risk of hemorrhagic complications, the risk of bleeding from other anatomical sites should also be considered when making this clinical decision.
In our study, there were 3 cases of bleeds due to SCI-related surgery (2.4%), with 1 requiring return to surgery (0.8%). Our reported bleed rates related to SCI surgery are within the lower range of previously reported bleeding complication rates (0%–9.5%). We did find a moderate rate of bleeding complications not related to SCI surgery (8.8%). It is important to note that each study may have different definitions of a bleeding complication. While many studies examining VTE rates in patients with SCI define bleeding complications as worsening intraspinal hematomas or bleeding requiring surgical intervention, very few consider other sites of hemorrhage such as bleeding due to tracheostomy tube placement, LD placement, PEG tube, or Foley catheter. To our knowledge, this is the first study to systematically categorize and examine different types of bleeding complications outside of intraspinal hematomas or life-threatening hemorrhage in patients with SCI who were administered chemoprophylaxis. As neurosurgical intervention is typically the limiting factor in chemical DVT prophylaxis administration, these non–SCI-related bleeding complications likely would have occurred regardless of our protocol.
Our study also found an association between SCI at the thoracic level and VTE, which is consistent with findings from previous studies, suggesting that the highest incidence of venous thromboembolic events occurs among patients with thoracic-segment SCI.2,8,10Similarly, the associations between ASIA grade and VTE, and lower-extremity motor score and VTE, have been shown in previous reports, suggesting that more severe paralysis is associated with higher rates of VTE.23,36Sharpe et al. observed that early operative fixation (within 3 days of injury) allowed for earlier patient mobilization and resulted in a lower incidence of complications such as pneumonia, shorter ICU LOS, and fewer ventilator days compared with late fixation.37Therefore, immobilization may increase the risk of VTE along with other complications, as noted in patients with worse lower-extremity motor scores and worse ASIA grades. Interestingly, age was found to be negatively associated with VTE in our study, which is in contrast to previous results in the literature.23,38,39Previous studies suggest that the higher rates of VTE in older adults may be due to lower activity levels, less robust cardiovascular and pulmonary functions, and more comorbidities.38It is also known that there are generally higher VTE incidences in the elderly.40It is possible that there may be other confounding variables that may moderate the association between age and VTE in our study. For example, younger patients may be more likely to undergo surgical intervention or have more severe injuries, both of which are risk factors for VTE. While we found an association between the race category of "other" and VTE, this is most likely a product of a small sample size. Although some studies do find associations between race/ethnicity and VTE,41this is a research question that would need to be further investigated. Overall, the usual predictors of VTE occurred with LMWH administration within 24 hours and therefore it remains critical to have patients undergo early surgery for earlier mobilization.
Limitations of the Study
There are several limitations associated with our study, including retrospective evaluation of prospectively collected data, which prevents exclusion of selection bias and introduces unevaluated differences as potential confounding variables. Additionally, this study lacks a control group due to our uniform VTE prophylaxis protocol across patients. Because VTEs were not ruled out in a systematic manner in all patients, potential detection bias may occur. Another limitation is the amount of missing data for certain variables, which prevented us from running further statistical analysis such as a multivariate regression. The majority of our missing data can be attributed to patients withdrawing from the study and the constraints that come with collecting certain data (such as motor scores and ASIA grade) in the hospital. The use of advanced predictive models in the future can help mitigate the effects of missing data on the analysis.
Conclusions
VTE following traumatic SCI is a major clinical concern. Utilizing the TRACK-SCI database, we found that a protocol for administration of LMWH within 24 hours of injury or surgery was associated with a low rate of spine surgery–related bleeding complications and a moderate rate of bleeding complications not related to spine surgery. Overall, these results suggest that earlier initiation of LMWH may be effective for VTE prophylaxis in patients with SCI, but the risk of low to moderate rates of bleeding that may be associated with early administration of LMWH should be considered. In this study, the authors did not consider administration of VTE prophylaxis earlier than 24 hours. However, given our findings, LMWH administration earlier than 24 hours may be warranted, especially in those with risk factors for VTE. A randomized controlled clinical trial would be safe to conduct in the future to better understand the optimal time to administer VTE prophylaxis along with its risks and benefits, including in cases with a higher risk of VTE.
Acknowledgments
Dr. Beattie received funding for this study from the Craig H. Neilsen Foundation, the US Department of Defense (grant nos. W81XWH-13-1-0297, W81XWH-16-1-0497, and SC190233), and Wings for Life United Kingdom (grant no. WFL-US-o7-18).
Disclosures
Dr. Ferguson reported receiving grants from the NIH/NINDS (nos. R01NS122888, UH3NS106899, and U24NS122732), Veterans Affairs (nos. I01RX002245, I01RX002787, I01BX0058771, and I50BX005878), the Wings for Life Foundation, and the Craig H. Neilsen Foundation during the conduct of the study. Dr. Mummaneni reported receiving personal fees from DePuy Synthes, Globus, NuVasive, Stryker, BK Medical, Brainlab, and SI Bone; book royalties from Thieme Publishing and Springer Publishing; grants from AO Spine, NIH/NIAMS (no. U19AR076737), the Patient-Centered Outcomes Research Institute (PCORI), Pacira Biosciences, the International Spine Study Group (ISSG), the Neurosurgery Research and Education Foundation (NREF), and Spinal Laminectomy Versus Instrumented Pedicle Screw Fusion II (SLIP II); and stock from Spinicity/ISD, outside the submitted work. Dr. Dhall reported receiving consulting fees from DePuy Synthes and royalties from Globus Medical outside the submitted work; in addition, Dr. Dhall had a patent for cerebrospinal cooling devices issued and a patent for a solid conduction hypothermia device issued. Dr. Saigal reported receiving personal fees from Proprio and Globus Medical, and nonfinancial support from Spiderwort outside the submitted work. Dr. Beattie reported being a principal investigator on a grant from the US Department of Defense (no. CDMRP SCIRP SC190233) outside the submitted work. Dr. DiGiorgio reported receiving grants from DePuy Synthes, the Mercatus Center at George Mason University, and AO Spine, and personal fees from the National Football League outside the submitted work.
Author Contributions
Conception and design: DiGiorgio, Chryssikos, Ferguson, Mummaneni, Duong-Fernandez, Singh. Acquisition of data: DiGiorgio, Lui, Chryssikos, Patel, Ferguson, Dhall, Duong-Fernandez, Saigal, Chou, Pan, Talbott, Pascual, Huie, Whetstone, Bresnahan, Beattie, Manley. Analysis and interpretation of data: DiGiorgio, Lui, Park, Chryssikos, Radabaugh, Patel, Aabedi, Dhall, Duong-Fernandez, Hemmerle, Kyritsis, Whetstone, Bresnahan, Weinstein. Drafting the article: DiGiorgio, Lui, Park, Chryssikos, Patel, Weinstein. Critically revising the article: DiGiorgio, Lui, Park, Chryssikos, Radabaugh, Ferguson, Mummaneni, Dhall, Duong-Fernandez, Saigal, Chou, Pan, Singh, Kyritsis, Talbott, Pascual, Whetstone, Beattie, Weinstein. Reviewed submitted version of manuscript: DiGiorgio, Lui, Park, Chryssikos, Radabaugh, Ferguson, Torres Espin, Mummaneni, Dhall, Saigal, Chou, Pan, Singh, Hemmerle, Kyritsis, Talbott, Pascual, Huie, Whetstone, Bresnahan, Beattie, Weinstein, Manley. Approved the final version of the manuscript on behalf of all authors: DiGiorgio. Statistical analysis: Lui, Chryssikos, Radabaugh, Ferguson, Weinstein. Administrative/technical/material support: DiGiorgio, Duong-Fernandez, Hemmerle, Pascual, Beattie, Manley. Study supervision: DiGiorgio, Chryssikos, Ferguson, Mummaneni, Duong-Fernandez, Beattie.
References
-
1 ↑
GeertsWH,CodeKI,JayRM,ChenE,SzalaiJP.A prospective study of venous thromboembolism after major trauma.N Engl J Med.1994;331(24):1601–1606.
-
2 ↑
RaksinPB,HarropJS,AndersonPA,et al.Congress of Neurological Surgeons systematic review and evidence-based guidelines on the evaluation and treatment of patients with thoracolumbar spine trauma: prophylaxis and treatment of thromboembolic events.开云体育app官方网站下载入口.2019;84(1):E39–E42.
-
3 ↑
GeertsWH,JayRM,CodeKI,et al.A comparison of low-dose heparin with low-molecular-weight heparin as prophylaxis against venous thromboembolism after major trauma.N Engl J Med.1996;335(10):701–707.
-
4
KnudsonMM,MorabitoD,PaiementGD,ShacklefordS.Use of low molecular weight heparin in preventing thromboembolism in trauma patients.J Trauma.1996;41(3):446–459.
-
5 ↑
CothrenCC,SmithWR,MooreEE,MorganSJ.Utility of once-daily dose of low-molecular-weight heparin to prevent venous thromboembolism in multisystem trauma patients.World J Surg.2007;31(1):98–104.
-
6 ↑
LouisSG,SatoM,GeraciT,et al.Correlation of missed doses of enoxaparin with increased incidence of deep vein thrombosis in trauma and general surgery patients.JAMA Surg.2014;149(4):365–370.
-
7 ↑
El MasriWS,SilverJR.Prophylactic anticoagulant therapy in patients with spinal cord injury.Paraplegia.1981;19(6):334–342.
-
8 ↑
JonesT,UgaldeV,FranksP,ZhouH,WhiteRH.Venous thromboembolism after spinal cord injury: incidence, time course, and associated risk factors in 16,240 adults and children.Arch Phys Med Rehabil.2005;86(12):2240–2247.
-
9 ↑
PloumisA,PonnappanRK,BesseyJT,PatelR,VaccaroAR.Thromboprophylaxis in spinal trauma surgery: consensus among spine trauma surgeons.Spine J.2009;9(7):530–536.
-
10 ↑
WorleyS,ShortC,PikeJ,AndersonD,DouglasJA,ThompsonK.Dalteparin vs low-dose unfractionated heparin for prophylaxis against clinically evident venous thromboembolism in acute traumatic spinal cord injury: a retrospective cohort study.J Spinal Cord Med.2008;31(4):379–387.
-
11
SlavikRS,ChanE,GormanSK,et al.Dalteparin versus enoxaparin for venous thromboembolism prophylaxis in acute spinal cord injury and major orthopedic trauma patients: ‘DETECT’ trial.J Trauma.2007;62(5):1075–1081.
-
12
Chiou-TanFY,GarzaH,ChanKT,et al.Comparison of dalteparin and enoxaparin for deep venous thrombosis prophylaxis in patients with spinal cord injury.Am J Phys Med Rehabil.2003;82(9):678–685.
-
13
ThumbikatP,PoonnoosePM,BalasubrahmaniamP,RavichandranG,McClellandMR.A comparison of heparin/warfarin and enoxaparin thromboprophylaxis in spinal cord injury: the Sheffield experience.Spinal Cord.2002;40(8):416–420.
-
14 ↑
Spinal Cord Injury Thromboprophylaxis Investigators. Prevention of venous thromboembolism in the acute treatment phase after spinal cord injury: a randomized, multicenter trial comparing low-dose heparin plus intermittent pneumatic compression with enoxaparin.J Trauma.2003;54(6):1116–1126.
-
15 ↑
ChangR,ScerboMH,SchmittKM,et al.Early chemoprophylaxis is associated with decreased venous thromboembolism risk without concomitant increase in intraspinal hematoma expansion after traumatic spinal cord injury.J Trauma Acute Care Surg.2017;83(6):1088–1094.
-
16
PloumisA,PonnappanRK,MaltenfortMG,et al.Thromboprophylaxis in patients with acute spinal injuries: an evidence-based analysis.中华骨科杂志.2009;91(11):2568–2576.
-
17 ↑
GreenD,LeeMY,LimAC,et al.Prevention of thromboembolism after spinal cord injury using low-molecular-weight heparin.Ann Intern Med.1990;113(8):571–574.
-
18 ↑
TracyBM,DunneJR,O’NealCM,ClaytonE.Venous thromboembolism prophylaxis in neurosurgical trauma patients.J Surg Res.2016;205(1):221–227.
-
19 ↑
FehlingsMG,TetreaultLA,AarabiB,et al.A clinical practice guideline for the management of patients with acute spinal cord injury: recommendations on the type and timing of anticoagulant thromboprophylaxis.Global Spine J.2017;7(3 suppl):212S–220S.
-
20 ↑
DhallSS,HadleyMN,AarabiB,et al.Deep venous thrombosis and thromboembolism in patients with cervical spinal cord injuries.开云体育app官方网站下载入口.2013;72(suppl 2):244–254.
-
21 ↑
ArnoldPM,HarropJS,MerliG,et al.Efficacy, safety, and timing of anticoagulant thromboprophylaxis for the prevention of venous thromboembolism in patients with acute spinal cord injury: a systematic review.Global Spine J.2017;7(3 suppl):138S–150S.
-
22 ↑
Consortium for Spinal Cord Medicine. Early acute management in adults with spinal cord injury: a clinical practice guideline for health-care professionals.J Spinal Cord Med.2008;31(4):403–479.
-
23 ↑
Prevention of Venous Thromboembolism in Individuals with Spinal Cord Injury: Clinical Practice Guidelines for Health Care Providers,.3rd ed.:Consortium for Spinal Cord Medicine.Top Spinal Cord Inj Rehabil.2016;22(3):209–240.
-
24 ↑
TsolinasRE,BurkeJF,DiGiorgioAM,et al.Transforming Research and Clinical Knowledge in Spinal Cord Injury (TRACK-SCI): an overview of initial enrollment and demographics.Neurosurg Focus.2020;48(5):E6.
-
25 ↑
HarrisPA,TaylorR,ThielkeR,PayneJ,GonzalezN,CondeJG.Research electronic data capture (REDCap)—a metadata-driven methodology and workflow process for providing translational research informatics support.J Biomed Inform.2009;42(2):377–381.
-
26 ↑
AitoS,PieriA,D’AndreaM,MarcelliF,CominelliE.Primary prevention of deep venous thrombosis and pulmonary embolism in acute spinal cord injured patients.Spinal Cord.2002;40(6):300–303.
-
27 ↑
GreenD,ChenD,ChmielJS,et al.Prevention of thromboembolism in spinal cord injury: role of low molecular weight heparin.Arch Phys Med Rehabil.1994;75(3):290–292.
-
28 ↑
MerliGJ,CrabbeS,DoyleL,DitunnoJF,HerbisionGJ.Mechanical plus pharmacological prophylaxis for deep vein thrombosis in acute spinal cord injury.Paraplegia.1992;30(8):558–562.
-
29 ↑
ZeeshanM,KhanM,O’KeeffeT,et al.Optimal timing of initiation of thromboprophylaxis in spine trauma managed operatively: a nationwide propensity-matched analysis of trauma quality improvement program.J Trauma Acute Care Surg.2018;85(2):387–392.
-
30 ↑
JacobsLJ,WoodsBI,ChenAF,LunardiniDJ,HohlJB,LeeJY.Safety of thromboembolic chemoprophylaxis in spinal trauma patients requiring surgical stabilization.Spine (Phila Pa 1976).2013;38(16):E1041–E1047.
-
31 ↑
DiGiorgioAM,TsolinasR,AlazzehM,et al.Safety and effectiveness of early chemical deep venous thrombosis prophylaxis after spinal cord injury: pilot prospective data.Neurosurg Focus.2017;43(5):E21.
-
32 ↑
AhlquistS,ParkHY,KelleyB,HollyL,ShamieAN,ParkDY.Venous thromboembolism chemoprophylaxis within 24 hours of surgery for spinal cord injury: is it safe and effective?Neurospine.2020;17(2):407–416.
-
33 ↑
ZhengR,GuanB,FanY,et al.A critical appraisal of clinical practice guidelines for management of four common complications after spinal cord injury.Spine J.2023;23(6):888–899.
-
34 ↑
RivasL,VellaM,JuT,et al.Early chemoprophylaxis against venous thromboembolism in patients with traumatic brain injury.Am Surg.2022;88(2):187–193.
-
35 ↑
FrisoliFA,ShinsekiM,NwabuobiL,et al.Early venous thromboembolism chemoprophylaxis after traumatic intracranial hemorrhage.开云体育app官方网站下载入口.2017;81(6):1016–1020.
-
36 ↑
MatsumotoS,SudaK,IimotoS,et al.Prospective study of deep vein thrombosis in patients with spinal cord injury not receiving anticoagulant therapy.Spinal Cord.2015;53(4):306–309.
-
37 ↑
SharpeJP,GobbellWC,CarterAM,et al.Impact of venous thromboembolism chemoprophylaxis on postoperative hemorrhage following operative stabilization of spine fractures.J Trauma Acute Care Surg.2017;83(6):1108–1113.
-
38 ↑
ChungWS,LinCL,ChangSN,ChungHA,SungFC,KaoCH.Increased risk of deep vein thrombosis and pulmonary thromboembolism in patients with spinal cord injury: a nationwide cohort prospective study.Thromb Res.2014;133(4):579–584.
-
39 ↑
Giorgi PierfranceschiM,DonadiniMP,DentaliF,et al.The short- and long-term risk of venous thromboembolism in patients with acute spinal cord injury: a prospective cohort study.Thromb Haemost.2013;109(1):34–38.
-
40 ↑
KniffinWDJr,BaronJA,BarrettJ,BirkmeyerJD,AndersonFAJr.The epidemiology of diagnosed pulmonary embolism and deep venous thrombosis in the elderly.Arch Intern Med.1994;154(8):861–866.
-
41 ↑
WhiteRH,KeenanCR.Effects of race and ethnicity on the incidence of venous thromboembolism.Thromb Res.2009;123(suppl 4):S11–S17.