Chronicsubdural hematoma (cSDH) is a growing public health problem in the United States, especially for the elderly. It is the most common intracranial hemorrhage among the elderly and will become the most common neurosurgical pathology by 2030.1,2The US Census reported that, by 2035, the older adult population is expected to outnumber children.3Furthermore, as people in the Western world live longer, there are more patients on antiplatelet and antithrombotic medications that exacerbate the bleeding risk.4cSDH is a concern for older adults because of the greater comorbidities that are associated with an advanced age. In a cohort study of a national Finnish database of patients with evacuated cSDH (n = 8539), the only statistically significant predictor of mortality by 1 year was old age.14Additionally, an age ≥ 55 years and male sex were the only significant predictors of reoperation for recurrent cSDH. A subsequent operation can increase the risk of mortality and morbidity in this aging population.2Understanding the risk factors associated with the need for a second operation for cSDH may reveal viable treatment pathways to avoid repeat surgery.
Prior studies have reported recurrence rates of 10%–24% following cSDH evacuation.5Given such a high incidence, a spectrum of prognostic models have been reported in the literature and internally validated. Among the most common predictors retained in the final models are older age, initial hematoma size and density, greater midline shift, and postoperative cavity volume.5–7However, only a few studies have examined the potential role of postsurgical complications, existing comorbidities,8statin or antithrombotic medication use,9or platelet count prior to the first evacuation, with equivocal results.10Given the longer life expectancy in the US, these variables will be of significant concern for the population moving forward.2Moreover, nearly all prior studies have used univariate and/or multivariate logistic regression analyses that have included dichotomous predictors to develop their models.5,6,11–18This method is commonly used in logistic regression analyses to enhance the interpretability of the association between predictor and outcome. For example, Suero Molina et al.13presented a prognostic model for cSDH recurrence that included preexisting thrombocytopenia as a significant predictor. Thrombocytopenia was documented as a binary variable, which may reduce the variability accounted for and miss potentially crucial information regarding viable cut points for intervention in this common preexisting condition.
The purpose of the present study was to leverage a data mining algorithm, chi-square automatic interaction detection (CHAID), which can incorporate continuous, nominal, and binary data into a decision tree, to identify the most robust predictors of repeat surgery for cSDH patients. We hypothesized that applying CHAID to a data set of cSDH patients with an augmented set of relevant predictors, as compared to those in the literature, will elucidate novel and compelling findings to extend prognostic modeling in this population.
开云体育世界杯赔率
Study Design and Participants
We performed a retrospective cohort study of all patients who had undergone cSDH evacuation performed by 15 neurosurgeons between 2011 and 2020. We included patients from two level 1 trauma centers at the University of Pittsburgh. To be included in the analysis, the subject must have presented with symptomology (e.g., headache, dizziness, imbalance, motor weakness) and had an associated cSDH detected on CT and requiring surgical intervention. We retrospectively reviewed patient charts for demographic and clinical information, as well as cSDH recurrence. By definition, a cSDH recurrence required a repeat surgery for cSDH evacuation within 1 year of the original treatment day, excluding surgeries for infection. The need for repeat surgery was determined on a case-by-case basis by the managing surgeon based on a patient’s functional and/or neurological status. We excluded patients with solely acute SDH and previous neurosurgical pathology (i.e., ventriculoperitoneal shunt, brain metastasis). This study was approved by the University of Pittsburgh Institutional Review Board for human subject research. Patient consent was provided to extract de-identified electronic health record information for analysis. Research procedures followed the ethical standards of the responsible committee on human experimentation and the Declaration of Helsinki 1975. De-identified data and modeling analysis are available to qualified researchers upon request.
Institutional Management of cSDH
The majority of neurosurgeons at our institution use a mini craniotomy for the treatment of cSDH. Typically, patients are placed under general anesthesia, and a small incision is made over the cranial convexity. A small, 3- to 4-cm-diameter mini craniotomy is made and copiously irrigated with warmed saline. Some neurosurgeons will use one or two burr holes at their discretion. The standard at our institution, and consistent with randomized controlled trials, is to place a subdural drain that remains in place until postoperative day 2 or 3.19Patients are mobilized immediately after surgery and are not confined to the bed.
The management of antithrombotic and antiplatelet agents was at the discretion of the managing neurosurgeon but typically followed guidelines from the Neurocritical Care Society.20If feasible, the effects of warfarin and direct-acting oral anticoagulants were allowed to wear off through waiting until either normalization of the international normalized ratio or 5 half-lives, respectively. Antiplatelet agents were managed similarly, with a 7-day waiting period for clopidogrel and 4-day waiting period for aspirin. In situations in which the cSDH threatened life or severe neurological injury, antiplatelets and anticoagulants were reversed according to guidelines. Anticoagulants and antiplatelets were restarted, if appropriate, typically after 1 month and a stable head CT scan. Agents were restarted sooner for more serious, life-threatening pathologies such as active pulmonary embolism or drug-eluting cardiac stents.
Data Analysis
We used descriptive statistics for the overall sample, as well as chi-square analyses and odds ratios to identify relevant univariate clinical predictors for cSDH recurrence. The CHAID algorithm is a data mining algorithm that we used to differentiate patients who required retreatment for cSDH from those who did not. The use of CHAID to develop prognostic models has several advantages over multivariable logistic regression. CHAID is a nonparametric statistical analysis that has no requirements to conform to the normal distribution assumptions of logistic regression modeling.21CHAID adjusts for multiple comparisons, which is a limitation of logistic regression. Furthermore, CHAID can automatically identify interactions between included predictors in the model, as well as significant relationships within nominal variables, and other included predictors.22CHAID can also identify the point in a continuous variable that maximizes the difference in the outcome (i.e., repeat cSDH surgery).23A decision tree output is produced, showing the identified predictors of the outcome in order of strength from top to bottom. The algorithm proceeds iteratively through the available predictors until the cohort cannot be further subdivided. Each node split is assessed with Bonferroni corrections for statistical significance. These CHAID characteristics are executed automatically by the algorithm based purely on the mathematical information provided in the data set.21The investigator does not select or control which cut point or combinations for each variable are included in the final model. The risk ratio and 95% confidence intervals are reported for each node split. For our model, we included the following predictors: 1) continuous: age, platelet count on admission, midline shift on admission, midline shift following the index cSDH evacuation, and greatest width of the preoperative hematoma on axial slices (in millimeters); 2) nominal: postsurgical complications after the first surgery (e.g., cerebrovascular accident, pneumonia, sepsis, deep vein thrombosis/pulmonary embolism, urinary tract infection, acute kidney injury), preexisting comorbidities (e.g., hypertension, abnormal liver function, heart failure/atrial fibrillation, chronic kidney disease, coronary artery disease, malignancy); 3) binary: biological sex at birth, statin use, anticoagulant use (i.e., warfarin, rivaroxaban, apixaban, enoxaparin), or antiplatelet use (i.e., aspirin or clopidogrel).
统计istical significance was set at p < 0.05. Statistics were completed using SPSS version 28.0.1 and SPSS Modeler version 18 (IBM Corp.).
Results
Demographics and Odds Ratios
Overall, 60 (13.8%) of 435 study-eligible patients had a cSDH recurrence. These patients had a larger SDH on presentation, lower platelet count, and higher odds of at least one postsurgical complication (Table 1). Patients with a cSDH recurrence also had 2.0 times greater odds of having used preoperative anticoagulants. There was no difference between patients who required a second intervention and those who did not in terms of age, number of males, use of preoperative antiplatelet medication, rate of comorbidities, or rate of postsurgical complications (Tables 1and2). One in 4 patients who required retreatment had been taking presurgical warfarin (Table 3). Nearly half of the patients requiring retreatment had been taking aspirin, and almost one-third had not been taking anticoagulants or antiplatelet medications.
的差异首先是表示信息en patients with repeat surgical intervention for SDH compared to those without
| Variable | Repeat Surgical Intervention | p Value | Cohen’s d* | OR | |
|---|---|---|---|---|---|
| Yes | No | ||||
| No. of patients | 60 | 375 | |||
| Age in yrs | 73.4 ± 11.1 | 74.0 ± 11.6 | 0.74 | 0.05 | — |
| 1st presentation | |||||
| ICU LOS in days | 2.6 ± 3.7 | 1.8 ± 2.5 | 0.06 | 0.29 | — |
| Midline shift in mm | 7.8 ± 4.6 | 6.9 ± 4.6 | 0.15 | 0.21 | — |
| Hematoma size in mm | 21.8 ± 6.7 | 18.3 ± 7.3 | 0.002 | 0.49 | — |
| Admission platelet count in × 109L | 180.7 ± 57.9 | 224.5 ± 82.9 | <0.001 | 0.55 | — |
| ≥1 comorbidity | 52 (86.7) | 291 (77.6) | 0.09 | — | 1.96 |
| ≥1 complication | 30 (50.0) | 106/373 (28.4) | 0.002 | — | 2.36 |
| 1st procedure | |||||
| 颅骨切开术 | 53 (88.3) | 312/373 (83.6) | 0.32 | — | 1.52 |
| 颅骨切开术w/ burr holes | 2 (3.3) | 0/373 (0) | <0.001 | — | 12.93 |
| Burr holes | 5 (8.3) | 61/373 (16.4) | 0.12 | — | 0.47 |
| 2nd procedure | |||||
| 颅骨切开术 | 46/59 (7.8) | — | — | — | — |
| Burr holes | 7/59 (11.9) | — | — | — | — |
| MMA embolization | 5/59 (8.5) | — | — | — | — |
| Other | 1/59 (1.7) | — | — | — | — |
— = not applicable; LOS = length of stay; MMA = middle meningeal artery.
Values are expressed as mean ± standard deviation or number (%), unless indicated otherwise. Boldface type indicates statistical significance (p < 0.05).
Effect size.
Prevalence of risk factors for repeated surgical intervention for SDH
| Variable | Repeat Surgical Intervention | OR | 95% CI | p Value | |
|---|---|---|---|---|---|
| Yes | No | ||||
| No. of patients | 60 | 375 | |||
| Male sex | 49 (81.7) | 265 (70.7) | 1.85 | 0.90–4.09 | 0.08 |
| Preop anticoagulant use | 19 (31.7) | 70 (18.7) | 2.01 | 1.10–3.68 | 0.02 |
| Preop antiplatelet use | 28 (46.7) | 181 (48.3) | 0.93 | 0.54–1.61 | 0.80 |
| Preop statin use | 34 (56.7) | 169 (45.1) | 1.55 | 0.88–2.74 | 0.13 |
| Comorbidities | |||||
| Hypertension | 35 (58.3) | 233 (62.1) | 0.85 | 0.47–1.55 | 0.57 |
| Abnormal liver function | 5 (8.3) | 14 (3.7) | 2.34 | 0.81–6.77 | 0.16 |
| Heart issue/atrial fibrillation | 15 (25.0) | 85 (22.7) | 1.14 | 0.60–2.14 | 0.69 |
| Malignancy | 12 (20.0) | 57 (15.2) | 1.40 | 0.70–2.79 | 0.35 |
| Chronic kidney disease | 4 (6.7) | 34 (9.1) | 0.72 | 0.25–2.20 | 0.54 |
| Coronary artery disease | 12 (20.0) | 81 (21.6) | 0.91 | 0.46–1.79 | 0.78 |
| Complications | |||||
| Cardiovascular accident | 3 (5.0) | 9 (2.4) | 2.14 | 0.56–8.14 | 0.22 |
| Sepsis | 2 (3.3) | 13 (3.5) | 0.96 | 0.21–4.37 | >0.99 |
| Pneumonia | 1 (1.7) | 14 (3.7) | 0.44 | 0.06–3.39 | 0.71 |
| Acute kidney injury | 2 (3.3) | 16 (4.3) | 0.77 | 0.17–3.45 | >0.99 |
| DVT/PE | 6 (10.0) | 17 (4.5) | 2.34 | 0.88–6.20 | 0.11 |
| Urinary tract infection | 6 (10.0) | 23 (6.1) | 1.70 | 0.66–4.37 | 0.27 |
DVT = deep vein thrombosis; PE = pulmonary embolism.
Values are expressed as number (%). Boldface type indicates statistical significance (p < 0.05).
Number of patients taking specific anticoagulant and antiplatelets and the rate of retreatment for cSDH
| Anticoagulant/Antiplatelet Agent | No. of Patients Taking Medication (n = 434) | Rate of Retreatment (n = 60) |
|---|---|---|
| Warfarin | 73 (16.8) | 15 (25.0) |
| Aspirin | 197 (45.4) | 27 (45.0) |
| Clopidogrel | 35 (8.1) | 2 (3.3) |
| Apixaban | 4 (0.9) | 2 (3.3) |
| Enoxaparin | 3 (0.7) | 1 (1.7) |
| Rivaroxaban | 8 (1.8) | 1 (1.7) |
| Multiple | 49 (11.3) | 7 (11.7) |
| None | 162 (37.3) | 18 (30.0) |
Values are expressed as number (%).
Differentiating Patients Requiring a Second Surgery for SDH From Those Who Did Not
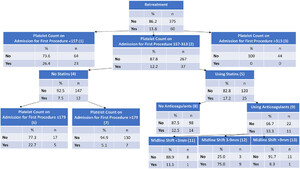
A CHAID decision tree model was built to differentiate patients with cSDH recurrence (Fig. 1). The final model had 13 nodes and 4 split levels. The model had an overall accuracy of 87.4% and an area under the curve of 0.76.

CHAID decision tree model. (number) = node identifier for statistical comparison.
Split Level 1
The predictor with the strongest association with cSDH recurrence was admission platelet count. Approximately 26% of patients (n = 23/87) with a platelet count < 157 × 109/L had a cSDH recurrence, whereas none of the 44 patients with a platelet count > 313 × 109/L had a cSDH recurrence. Patients between that range (157–313 × 109/ L;n = 12.2 304)需要一个过程% rate (n = 37). A dose-response relationship was observed between a lower platelet count on first admission and an increased risk of recurrence (Table 4).
Risk ratios between predictor nodes from the CHAID decision tree
| Node Comparison | RR | 95% CI | p Value |
|---|---|---|---|
| Platelets <157 vs >313 × 109/L | 11.90 | 1.66–85.26 | 0.001 |
| Platelets 157–313 vs >313 × 109/L | 5.48 | 0.77–38.94 | 0.046 |
| Platelets <157 vs 157–313 × 109/L | 2.17 | 1.35–3.47 | 0.001 |
| 统计in use vs no statin use | 2.28 | 1.19–4.38 | 0.01 |
| Platelets ≤179 vs 179 × 109/L | 4.45 | 1.55–12.78 | 0.004 |
| Anticoagulant use vs no anticoagulant use | 2.67 | 1.34–5.34 | 0.01 |
| Midline shift 3–9 vs <3 mm | 6.75 | 1.03–44.08 | 0.004 |
| Midline shift 3–9 vs >9 mm | 9.00 | 1.34–60.46 | 0.001 |
Boldface type indicates statistical significance (p < 0.05).
Split Level 2
The next strongest predictor of the risk for a second procedure was preoperative statin use among the patient group with 157–313 × 109/L platelet count. Approximately 17% of patients in this category who had used statins required a second procedure, which was associated with a 2.3 times greater risk for repeat surgery compared to patients not using statins preoperatively.
Split Level 3
Among those who had not used preoperative statins, a platelet count ≤ 179 × 109/L on admission for the first procedure was the strongest predictor of a second surgery. Nearly 23% (n = 5/22) of patients in this category required a second procedure, which was associated with a 4.5 times increased risk compared to patients with a platelet count > 179 × 109/L and not using statins.
Among those who had used preoperative statins, the use of preoperative anticoagulants was the strongest predictor of repeat surgery. Approximately 33% (n = 11/33) of patients using statins and anticoagulants required a second surgery, which was associated with a 2.7 times greater risk compared to patients using statins but not anticoagulants.
Split Level 4
The final indicator of patients requiring a second procedure was midline shift. Among patients using preoperative statins and anticoagulants, a midline shift between 3–9 mm required a second surgery at a 75% rate (n = 9/12). This shift was associated with a 6.8 times increased risk for a second surgery compared to patients with a midline shift < 3 mm and a 9.0 times greater risk for a second surgery compared to patients with a midline shift > 9 mm.
Discussion
We used CHAID, a decision tree algorithm, to develop a prognostic model of cSDH recurrence and identified platelet count, statin and/or anticoagulant use, and midline shift as significant predictors. The most important driver of the risk of cSDH recurrence was admission platelet count. Patients with platelets below a 157 × 109/L threshold had a 26% prevalence for cSDH recurrence, compared to 11% for those above that threshold. None of the 44 patients with a high platelet count (> 313 × 109/L) had a cSDH recurrence. The next strongest predictor of cSDH recurrence was preoperative statin use, an agent with known antiplatelet effects. The use of anticoagulants in addition to statins further exacerbated the cSDH recurrence risk by an additional 167% among patients who presented with platelet counts of 157–313 × 109/L. Our study extends prior work on prognostic modeling for recurrent cSDH surgery through identifying several, clinically relevant, population-specific cut points that increase the risk of cSDH recurrence.
The major strength of our study is that many of the cutoff points we identified may be clinically modifiable. While previously reported age and preoperative cSDH volumes are known predictors of cSDH recurrence,5they are not modifiable clinical characteristics. In contrast, we identified three groups of patients based on preoperative platelet count (group 1: < 157 × 109/L, group 2: 157–313 × 109/L, group 3: > 313 × 109/L) that have large differences in cSDH recurrence (26.4%, 12.2%, and 0%, respectively). Without significant supporting evidence, traditional neurosurgery dogma teaches that patients with < 100,000 platelets have an increased risk for postoperative hematoma.4我们的研究结果表明,更高的阈值(即,157,000) may be necessary to reduce the risk of cSDH recurrence. Importantly, our work supports only a correlative, not a causative, relationship between admission platelet count and cSDH recurrence. A low platelet count can reflect poor underlying health, such as alcoholic cirrhosis or chronic kidney disease. While we attempted to control for underlying preoperative health conditions (i.e., chronic kidney disease, abnormal liver function), future studies should assess whether higher preoperative platelet goals reduce the postoperative hemorrhage risk.
In a vein similar to the platelets, anticoagulation and antithrombic agents increased the risk of cSDH recurrence within a subgroup of our cohort that presented with a platelet count of 157–313 × 109/L. Our institution follows the Neurocritical Care Society guidelines for the management of these agents and waits for these agents to wear off unless severe life or neurological risks preclude waiting.20For emergency surgeries, we reversed the effects with appropriate blood products or reversal agents. Despite this, their preoperative use still increased the risk for cSDH recurrence. Likewise, these agents also pose a risk when restarting them after surgery. Patients are screened for unnecessary antiplatelet and antithrombotic agents, and those without a clear indication are stopped (e.g., stop aspirin in a patient with falls who takes it for "heart health"). For patients who need restarting of antiplatelet and anticoagulation medications, we try to wait a full month prior to restart with a normal head CT. Despite this conservative, data-driven approach, these agents pose significant risk for cSDH recurrence, and patients should be counseled about their risks.
The CHAID model also revealed that preoperative statin use, a medication with significant antiplatelet effects, is correlated with an increased risk for cSDH recurrence among those patients who present with a platelet count of 157–313 × 109/L.24Postoperatively, statins appear to have a beneficial effect of cSDH reabsorption, likely through modifying inflammatory pathways in cSDH formation. A prior umbrella study of systematic reviews and meta-analyses of cSDH recurrence found similar results to those obtained with the CHAIN model.10Most of this effect is driven by patients who are on statins and an anticoagulant, for whom the risk of recurrence is 2.7 times higher compared to those not on an anticoagulant. While our results do not prove causality, the increased risk may be related to an antiplatelet effect for statins that reduced both the quantity and effectiveness of platelets.
A similar effect has been found in patients with intracerebral hemorrhage (ICH) from stroke.25,26While statins improve functional outcomes if initiated after ICH,27pre-ICH使用可以增加spont的风险aneous ICH, possibly due to antiplatelet effects. Although postoperative statin use, through its antiinflammatory responses, has been proven beneficial at reducing cSDH volume, little is known about the preoperative risks of statin use. Future studies should evaluate whether preoperative platelet transfusions reduce the risks of cSDH recurrence for patients on statins preoperatively. More evidence will be necessary to understand if the use of statins and/or anticoagulants is a reversible risk factor for cSDH recurrence. Future research should also investigate whether the role of statins and/or anticoagulants is only relevant to recurrence when presenting with a certain platelet count, which evidence from this study would suggest.
One of the most common predictors included in previous prognostic models for recurrent cSDH surgery is older age.5,18,28Although age was included as a predictor in our models, it was not retained in the final decision tree. This may indicate that older age is a proxy variable for more specific predictors, such as lower platelet count and the use of statins/anticoagulant medications. Platelet count decreases with age, with lower average values observed in males.29Similarly, older adults are more likely to be taking statins and anticoagulant medications than younger adults because of the higher risk of comorbidities like stroke, atrial fibrillation, and heart disease.30,31Our results suggest that chronological and biological age—that is, the difference between evaluating age as a number and evaluating it as the cumulative effect of disease and aging on the body—may have differing effects on the risk of cSDH recurrence.
Study Limitations
Our work has several limitations. Our data set did not include some relevant predictors from prior work, including pre- and postoperative hematoma volume. Several other groups have shown that larger hematoma volumes are associated with an increased risk for cSDH recurrence.11,12委员会宽度,而在我们的研究中,使用爵士ves as a proxy for volume, hematoma volume itself may be more predictive. Additionally, our model was developed with data from a single institution. While we included patients from two level 1 trauma centers covered by 15 different neurosurgeons, external validation is needed before widespread use of our model. We did not document the neurosurgeon who performed each procedure, so a comparison of results across surgeons was not possible. The model presented here has only been internally validated and requires validation in external data sets. Future work should directly compare the performance of the model presented here with other prognostic models reported in the literature. Lastly, we collapsed all complications into one binary variable to simplify modeling efforts. This results in a loss of variance in certain outcomes, such as the difference in severity of some postoperative complications.
Conclusions
We applied a data mining decision tree algorithm to a retrospectively reviewed data set of cSDH patients to uncover novel insights for predicting cSDH recurrence. Our CHAID model identified platelet count on admission as the most important predictor of repeat cSDH surgery, followed by preoperative statin use and anticoagulant use. Notably, the decision tree model identified critical cutoffs for platelet count, which future studies should assess as modifiable or reflective of underlying disease states. Within a cohort that presented to the hospital with a platelet count of 157–313 × 109/L, preoperative statin and anticoagulant use further increased the risk for cSDH recurrence.
Disclosures
Dr. Vargas reported stock options with Viz.ai; consulting for Imperative Care, Scientia, Q’Apel, Medtronic, Cerenovus; and equity in Synchron and Borvo; all outside the submitted work.
Author Contributions
Conception and design: Eagle, Kellogg, Nwachuku, Okonkwo, Pease. Acquisition of data: Mittal, Nwachuku, Deng, Okonkwo, Pease. Analysis and interpretation of data: Eagle, Mittal, Deng, Okonkwo, Pease. Drafting the article: Eagle, Nwachuku, Deng, Okonkwo, Pease. Critically revising the article: Eagle, Vargas, Nwachuku, Deng, Buell, Okonkwo, Pease. Reviewed submitted version of manuscript: Eagle, Mittal, Kellogg, Vargas, Buell, Okonkwo, Pease. Approved the final version of the manuscript on behalf of all authors: Eagle. Statistical analysis: Eagle. Study supervision: Okonkwo.
References
-
1 ↑
ShapeyJ,GlanczLJ,BrennanPM.Chronic subdural haematoma in the elderly: is it time for a new paradigm in management?Curr Geriatr Rep.2016;5(2):71–77.
-
2 ↑
WeigelR,SchillingL,KraussJK.The pathophysiology of chronic subdural hematoma revisited: emphasis on aging processes as key factor.Geroscience.2022;44(3):1353–1371.
-
3 ↑
VespaJ.The graying of America: more older adults than kids by 2035.United States Census Bureau.Published March 13, 2018. Accessed August 4, 2023.https://www.census.gov/library/stories/2018/03/graying-america.html
-
4 ↑
LiD,GlorT,JonesGA.Thrombocytopenia and neurosurgery: a literature review.World Neurosurg.2017;106:277–280.
-
5 ↑
HollDC,MikolicA,BlaauwJ,et al.External validation of prognostic models predicting outcome after chronic subdural hematoma.Acta Neurochir (Wien).2022;164(10):2719–2730.
-
6 ↑
ZhangJJY,WangS,喷火ASC,et al.Development of a prognostic scoring system to predict risk of reoperation for contralateral hematoma growth after unilateral evacuation of bilateral chronic subdural hematoma.J Clin Neurosci.2020;78:79–85.
-
7 ↑
YuW,ChenW,JiangY,et al.Effectiveness comparisons of drug therapy on chronic subdural hematoma recurrence: a Bayesian network meta-analysis and systematic review.Front Pharmacol.2022;13:845386.
-
8 ↑
Martinez-PerezR,TsimpasA,RayoN,CepedaS,LagaresA.Role of the patient comorbidity in the recurrence of chronic subdural hematomas.Neurosurg Rev.2021;44(2):971–976.
-
9 ↑
XuM,ChenP,ZhuX,WangC,ShiX,YuB.Effects of atorvastatin on conservative and surgical treatments of chronic subdural hematoma in patients.World Neurosurg.2016;91:23–28.
-
10 ↑
ZhuF,WangH,LiW,et al.Factors correlated with the postoperative recurrence of chronic subdural hematoma: an umbrella study of systematic reviews and meta-analyses.EClinicalMedicine.2021;43:101234.
-
11 ↑
BartekJJr,SjåvikK,KristianssonH,et al.Predictors of recurrence and complications after chronic subdural hematoma surgery: a population-based study.World Neurosurg.2017;106:609–614.
-
12 ↑
HamouH,AlzaiyaniM,PjontekR,et al.Risk factors of recurrence in chronic subdural hematoma and a proposed extended classification of internal architecture as a predictor of recurrence.Neurosurg Rev.2022;45(4):2777–2786.
-
13 ↑
Suero MolinaE,BorscheidL,FreistühlerM,Zawy AlsofyS,StummerW,SchipmannS.风险评估慢性硬脑膜下血肿的国际化程度ated in 148 patients—a score for predicting recurrence.Clin Neurol Neurosurg.2020;195:106020.
-
14 ↑
PostiJP,LuotoTM,SipiläJOT,RautavaP,KytöV.Prognosis of patients with operated chronic subdural hematoma.Sci Rep.2022;12(1):7020.
-
15
ShenJ,GaoY,LiQ,et al.Risk factors predicting recurrence of bilateral chronic subdural hematomas after initial bilateral evacuation.World Neurosurg.2019;130:e133–e139.
-
16
StanišicM,PrippAH.A reliable grading system for prediction of chronic subdural hematoma recurrence requiring reoperation after initial burr-hole surgery.开云体育app官方网站下载入口.2017;81(5):752–760.
-
17
WonSY,DubinskiD,EibachM,et al.External validation and modification of the Oslo grading system for prediction of postoperative recurrence of chronic subdural hematoma.Neurosurg Rev.2021;44(2):961–970.
-
18 ↑
YanC,YangMF,HuangYW.A reliable nomogram model to predict the recurrence of chronic subdural hematoma after burr hole surgery.World Neurosurg.2018;118:e356–e366.
-
19 ↑
SantariusT,KirkpatrickPJ,GanesanD,et al.Use of drains versus no drains after burr-hole evacuation of chronic subdural haematoma: a randomised controlled trial.Lancet.2009;374(9695):1067–1073.
-
20 ↑
FronteraJA,LewinJJIII,RabinsteinAA,et al.Guideline for reversal of antithrombotics in intracranial hemorrhage: a statement for healthcare professionals from the Neurocritical Care Society and Society of Critical Care Medicine.Neurocrit Care.2016;24(1):6–46.
-
21 ↑
McCartyJA,HastakM.Segmentation approaches in data-mining: a comparison of RFM, CHAID, and logistic regression.J Bus Res.2007;60(6):656–662.
-
22 ↑
KassGV.An exploratory technique for investigating large quantities of categorical data.J R Stat Soc Ser C Appl Stat.1980;29(2):119–127.
-
23 ↑
ConnaboyC,EagleSR,JohnsonCD,FlanaganSD,MiQI,NindlBC.Using machine learning to predict lower-extremity injury in US Special Forces.Med Sci Sports Exerc.2019;51(5):1073–1079.
-
24 ↑
VioliF,CalvieriC,FerroD,PignatelliP.统计ins as antithrombotic drugs.Circulation.2013;127(2):251–257.
-
25 ↑
FitzMauriceE,WendellL,SniderR,et al.Effect of statins on intracerebral hemorrhage outcome and recurrence.Stroke.2008;39(7):2151–2154.
-
26 ↑
Sanz-CuestaBE,SaverJL.Lipid-lowering therapy and hemorrhagic stroke risk: comparative meta-analysis of statins and PCSK9 inhibitors.Stroke.2021;52(10):3142–3150.
-
27 ↑
DoerrfussJI,Abdul-RahimAH,SiegerinkB,et al.Early in-hospital exposure to statins and outcome after intracerebral haemorrhage—results from the Virtual International Stroke Trials Archive.Eur Stroke J.2020;5(1):85–93.
-
28 ↑
JackA,O’KellyC,McDougallC,FindlayJM.Predicting recurrence after chronic subdural haematoma drainage.Can J Neurol Sci.2015;42(1):34–39.
-
30 ↑
EzekowitzMD,FalkRH.The increasing need for anticoagulant therapy to prevent stroke in patients with atrial fibrillation.Mayo Clin Proc.2004;79(7):904–913.
-
31 ↑
NannaMG,NavarAM,WangTY,et al.统计in use and adverse effects among adults> 75 years of age: insights from the patient and Provider Assessment of Lipid Management (PALM) registry.J Am Heart Assoc.2018;7(10):e008546.