Oncedescribed as a surgical “no man’s land,”1the petroclival region remains one of the most complex skull base territories to approach surgically. This narrow space—between the medialpetrous apexlaterally, lower two-thirds of theclivusmedially, and brainstem posteriorly—is the crossroads for half of the cranial nerves (CNs) en route to their extradural destinations. Similarly, the majority of the posterior circulation vasculature traverses this region. Petroclival meningioma (PCM) arises at the petrous tip, medial to the internal auditory meatus and posterior to the gasserian ganglion.2These tumors may involve the upper clivus,cavernous sinus, Meckel’s cave,tentorium, and petrous apex to variable extents,3but large meningiomas inevitably involve the suprasellar, interpeduncular, prepontine, and ambient cisterns.4Identifying the best surgical approach for treating these lesions remains a challenge.
The combined petrosal (CP) approach, popularized by Hakuba et al.,5Al-Mefty et al.,2,6–8and many others,9,10has been used to resect PCMs. The CP approach combines the anterior transpetrosal approach with the retrolabyrinthine presigmoid transtentorial approach. Gross-total resection can be achieved in some patients with large PCMs while hearing andfacial nervefunction are preserved.8The CP approach also affords access to the posterior cavernous sinus, Meckel’s cave, posterolateral brainstem, and cerebellopontine angle, which facilitates tumor resection even in patients with firm lesions.8Nonetheless, the inherent morbidities associated with sigmoid sinus thrombosis, venous infarction,temporal lobeinjury secondary to excessive retraction, and damage to thelabyrinthmay dissuade surgeons and patients alike from choosing this approach.11,12
In our practice, we use the CP approach to resect large PCMs. Recently, however, a more anterolateral trajectory that combines the pretemporal transcavernous approach pioneered by Dolenc,13the anterior transpetrosal approach of Kawase et al.,14and trans–Meckel’s cave transtentorial exposure has emerged as an alternative for resection of large PCMs.4,15In a recent series by Liao et al.,4this pretemporal transcavernous anterior petrosal (PTAP) approach was used to achieve gross- or near-total resection of large PCMs with acceptable morbidity and no deaths.
Although both the CP and PTAP approaches may be feasible surgical options for managing PCMs, the indications for using one over the other remain unclear. Furthermore, objective data comparing both approaches are nonexistent. In this study, we conducted an anatomical investigation to 1) compare the technical nuances of the CP and the PTAP approaches, 2) provide quantitative comparisons of the exposures obtained with both approaches, and 3) identify key elements necessary to select the appropriate approach to PCMs.
开云体育世界杯赔率
The study was conducted in the surgical neuroanatomy laboratory at Barrow Neurological Institute. Twelve silicone-injected cadaveric head specimens were dissected using previously described methods.
CP Approach
VIDEO 1.手术视频说明圣的关键eps involved in performing the CP and PTAP approaches. Copyright Samy Youssef. Published with permission. Clickhereto view.

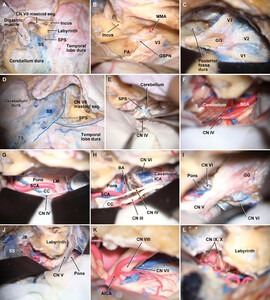
Photographs of stepwise cadaveric dissection illustrating the CP approach.A:Elevation of the sternomastoid–temporal fascia flap on the left side. The temporalis muscle is subsequently incised and reflected anteroinferiorly (not shown). The sigmoid sinus (SS) is fully skeletonized, and the jugular bulb (JB) is exposed. The antrum is opened, and the semicircular canals are skeletonized. The mastoid segment of the facial artery (CN VII mastoid seg.) is identified. Trautmann’s triangle dura (bound by the SS, SPS, otic capsule, and JB) is exposed. Subsequently, a kidney-shaped craniotomy is drilled, exposing the temporal, inferior parietal, and retrosigmoid dura.B:The middle meningeal artery (MMA) is divided to allow for further anterior exposure. The inferior aspect of V3 and the gasserian ganglion (GG) are also dissected off the underlying bone to maximize exposure of the petrous apex (PA).C:Anterior petrosectomy is performed to expose the dura overlying the pons immediately underneath the trigeminal nerve (CN V) root as it enters Meckel’s cave. Meckel’s cave is opened, and the GG is exposed.D:Theblue lineindicates the dural incision pattern used in this approach. The middle fossa dura at the base of the temporal lobe is incised from anterior to posterior, parallel to the SPS and as far back as possible above the transverse sinus. The presigmoid dura is incised in a vertical fashion, behind the endolymphatic sac, from the bulb up to the SPS where the incision curves anteriorly below the SPS. The temporal and presigmoid incisions are then joined, while the SPS is divided medial to where the superior cerebellar venous complex enters and drains into the SPS.E:The tentorium is divided while the trochlear nerve (CN IV) is preserved.F:The divided tentorium is retracted anteriorly and then resected. CN IV and the SCA are shown coursing through the posterior part of the ambient cistern.G:The lateral aspect of the lower crus cerebri (CC) and upper pons is exposed in a subtemporal fashion after resection of the tentorium.H:The posterior clinoid process has been drilled and the posterior cavernous sinus has been opened, exposing the cavernous segments of the ICA and abducens nerve (CN VI). This allows for greater exposure of the ipsilateral pons and for exposure of a longer segment of the basilar artery (BA).I:Magnified view showing both the cisternal and cavernous segments of CN VI.J:View obtained from the presigmoid window. This unobstructed, superficial view of the surgical field, below and above the root entry zone of the CN V, can be obtained only by division of the SPS and retraction of the transverse-sigmoid venous complex posteriorly.K:The presigmoid view also allows excellent visualization of the facial-cochlear complex.L:A small segment of the lower CNs is also visualized. AICA = anterior inferior cerebellar artery; LM = Liliequist membrane; TS = transverse sinus. Used with permission from Barrow Neurological Institute, Phoenix, Arizona.
Step 1: Elevation of the Sternomastoid–Temporal Fascia Flap
A C-shaped skin incision is made, extending from the level of themastoid tip, curving superiorly parallel to thesuperior temporal line, and then descending to the level of thezygomaapproximately two finger-widths anterior to theexternal auditory canal. The skin flap is retracted anteriorly and inferiorly. Thetemporalis fasciais incised and dissected posteriorly and in continuation with the sternomastoid muscle. Thetemporalis muscleis then incised and reflected anteroinferiorly.
Step 2: Retrolabyrinthine Presigmoid Exposure
The sigmoid sinus is fully skeletonized from its junction with thetransverse sinusdown to thejugular bulb. The antrum is opened, and the semicircular canals are exposed. The inferior aspect of the mastoid is drilled to expose the bulb. At the conclusion of this step, the dura mater of Trautmann’s triangle (bound by the sigmoid and superior petrosal sinuses, otic capsule, and jugular bulb) is exposed.
Step 3: Temporal, Parietal, Occipital Craniotomy
The dura overlying the sigmoid transverse junction is dissected. A kidney-shaped craniotomy exposing the temporal, inferior parietal, and lateral occipital (retrosigmoid) dura is fashioned.
Step 4: Anterior Petrosectomy
The dura of the temporal lobe is elevated off themiddle fossafloor from posterior to anterior. The greater superior petrosal nerve (GSPN) is identified and followed anteriorly as it intersectsV3at theforamen ovale. The inferior aspect of V3 and the gasserian ganglion are also dissected off the underlying bone and reflected as anterior as possible to maximize exposure of the petrous apex. In this study, we also continued extradural dissection as far anterior as possible to exposeV2and the posterior aspect of the cavernous sinus. The Kawase triangle—delineated by thearcuate eminence, GSPN, V3, and petrous ridge—is outlined and drilled.
Step 5: Dural Opening and Tentorium Resection
The dura overlying the base of the temporal lobe is incised from anterior to posterior, parallel to thesuperior petrosal sinus(SPS), and as far back as possible above the transverse sinus. The presigmoid dura is incised in a vertical fashion, behind the endolymphatic sac, from the bulb to the SPS, where the incision curves anteriorly below the SPS. The temporal and presigmoid incisions are then joined, while the SPS is divided medial to where the superior cerebellar venous complex enters and drains into the SPS. The tentorium is divided while thetrochlear nerve是保存了下来。硬脑膜形成Meckel的屋顶的s cave is cut, as well as the SPS above the porous, and the incision continues medially to include the tentorium; this maneuver completes the anterior cut of the tentorium resection. The posterior petroclinoid fold is divided, and the posterior aspect of the infratrochlear triangle is accessed.
PTAP Approach
The PTAP approach has been described elsewhere4,18and is summarized below (Fig. 2andVideo 1).

Photographs of stepwise cadaveric dissection illustrating the PTAP approach.A:Surgical view after completion of right-side orbitozygomatic craniotomy. The meningio-orbital fold (arrow) tethers the frontotemporal basal dura to the periorbita (PO).B:The meningio-orbital fold is divided as the outer dural layer is mobilized from anterior to posterior until the ACP is completely exposed and then drilled.C:Extradural dissection is continued posteriorly to expose V3 and the GSPN. The GG is completely exposed as peeling continues medially toward the tentorium.D:The PA segment to be drilled and the arcuate eminence (AE) are exposed.E:The PA is drilled, exposing the dura (asterisk) overlying the pons.F:The subtemporal dura, as well as that overlying the PA, has been opened. Similarly, the cavernous sinus has been opened, exposing CN III, CN IV, V1, and CN VI coursing below Gruber’s ligament (GL).G:The window afforded by drilling the PA and a part of the internal auditory canal.H:Magnified view of the nerves of the internal auditory canal. FD = frontal dura; P. maj. = portio major of the trigeminal nerve; P. min. = portio minor of trigeminal nerve; TD = temporal dura. SeeFig. 1for definitions of abbreviations. Used with permission from Barrow Neurological Institute, Phoenix, Arizona.
Step 1: Orbitozygomatic Craniotomy
An orbitozygomatic craniotomy is performed, and thelesser wing of the sphenoidbone is drilled medially toward the superiorsuperior orbital fissure.
Step 2: Exposure and Drilling of the Anterior Clinoid Process
The meningio-orbital fold is identified and divided. The meningio-orbital fold represents the easiest point at which to start dissection between the meningeal dura of the temporal lobe and the lateral wall of the cavernous sinus. The outer meningeal layer is mobilized from anterior to posterior until theanterior clinoid process(ACP) is completely exposed. Drilling begins at the superolateral aspect of theoptic canaland progresses medially. The core of the ACP is drilled up to the periosteal layer encasing theoptic nerve. Theoptic strutis then carefully drilled while great care is taken to avoid injury to the paraclinoidinternal carotid artery(ICA), which courses against the posterior surface of the strut.
Step 3: Anterior Petrosectomy
Extradural dissection is continued posteriorly to expose V3, ligate the middle meningeal artery, and expose the GSPN. The gasserian ganglion is completely exposed as peeling continues medially toward the tentorium. The Kawase triangle is exposed and drilled.
Step 4: Dural Opening and Tentorium Resection
A T-shaped dural opening is made with the vertical limb along thesylvian fissureand the horizontal limb, extending from the ACP along the base of the temporal lobe toward the site for anterior petrosectomy. Meckel’s cave is then opened over the gasserian ganglion toward the tentorial incisura.
Step 5: Combined Intradural-Extradural Mobilization of Neurovascular Structures
Multiple neurovascular elements are mobilized to create a safe corridor for drilling the posterior clinoid anddorsum sella. The falciform ligament and the distal dural ring are divided to facilitate ICA mobilization. Subsequently, the oculomotor trigone is opened, and the nerve is sharply mobilized up to its entrance to the superior orbital fissure while the adjacent trochlear nerve is protected. The proximal sylvian fissure is then opened widely, and the posterior clinoidal process (PCP) is exposed and drilled.
Quantitative Measurements
Measurements were obtained using the StealthStation stereotactic navigation system (Medtronic). The CP approach was performed in 5 specimens (group A), whereas the PTAP approach was performed in another 5 specimens (group B).Figure 3depicts the anatomical elements that were measured and compared.
Area of Drilled Clivus
Rhoton’s definition of the three clival segments was adapted.19,20因此,上部三分之一的斜坡延伸from the dorsum sella rostrally to a transverse line connecting to the dural pori of the abducens nerves caudally. The middle clivus extends below the upper clivus down to the level of the glossopharyngeal meatus, whereas the inferior limit of the lower clivus is represented by the lower border of the anterior aspect of the foramen magnum. Several coordinate points along the margins of the drilled region of the clivus were recorded, and the areas were calculated.
Neurovascular Elements and Brainstem
The lengths of bilaterally exposed CNs II–X, posterior cerebral artery,superior cerebellar artery(SCA), andvertebral artery, as well as the length of thebasilar artery, were recorded and compared between the two groups.
Several points on the ventral surface of the brainstem bilaterally were recorded: 1) superior-lateral exposable point on the ipsilateral side; 2) inferior-lateral exposable point on the ipsilateral side; 3) superior exposable point along a vertical line passing ipsilateral CNIII; 4) inferior exposable point along a vertical line passing ipsilateral CN III; 5) superior exposable point along the midline; 6) inferior exposable point along the midline; 7) superior-lateral exposable point on the contralateral side; 8) inferior-lateral exposable point on the contralateral side; 9) superior-lateral exposable point through the presigmoid window; 10) inferior-lateral exposable point through the presigmoid window; 11) inferior-medial exposable point through the presigmoid window; and 12) superior-medial exposable point through the presigmoid window (Fig. 3). The areas created on the surface of the brainstem were calculated and compared between the two groups.

A and B:Schematic illustrations showing ventral (A) and lateral (B) views of the brainstem and relevant neurovascular elements that were identified and measured. The following elements are shown: superior-lateral exposable point on the ipsilateral side (1); inferior-lateral exposable point on the ipsilateral side (2); superior exposable point along thevertical dashed linepassing ipsilateral CN III (3); inferior exposable point along thevertical dashed linepassing ipsilateral CN III (4); superior exposable point along the midline (5); inferior exposable point along the midline (6); superior-lateral exposable point on the contralateral side (7); inferior-lateral exposable point on the contralateral side (8); superior-lateral exposable point through the presigmoid window (9); inferior-lateral exposable point through the presigmoid window (10); inferior-medial exposable point through the presigmoid window (11); and superior-medial exposable point through the presigmoid window (12).C:Photograph of cadaveric dissection showing the anterior and posterior angles of attack, with the target being the abducens nerve as it crosses Gruber’s ligament. The anterior and posterior angles are outlined inredandblue, respectively. Used with permission from Barrow Neurological Institute, Phoenix, Arizona.
Angle of Attack
Maneuverability with each approach was evaluated with respect to a constant and easily accessible target, the point of intersection between CNVIand Gruber’s ligament. This value was quantitatively measured as the maximum allowable angle of attack in the horizontal plane. The angle of attack was measured by moving the proximal end of a probe from an extremely anterior position to an extremely posterior position, with the distal end placed at the target. A line perpendicular to the greater superficial petrosal nerve was defined as a neutral reference line, and the allowable angles of attack anterior and posterior to this line were defined as the anterior and posterior angles of attack, respectively. The total, anterior, and posterior angles of attack were measured and compared between the two approaches.
Statistical Analysis
Statistical comparisons were performed using PASW Statistics version 18.0.0 (IBM Corp.). Heron’s formula was used to calculate areas as the sum of triangles. The independent t-test was used for comparisons, and p < 0.05 was considered statistically significant.
Results
Morphometric Comparison of the CP and PTAP Approaches
Area of Drilled Clivus
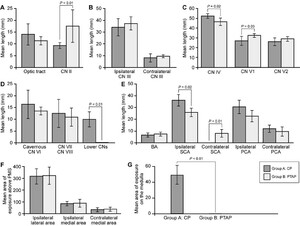
The predrilled areas of the upper third of the clivus were similar between the two groups (p = 0.60), but a significantly greater area was drilled in group A than group B (mean ± SD 88.7 ± 17.1 mm2vs 48.4 ± 17.9 mm2, p < 0.01)。The lower third of the clivus could not be exposed in group B.
Neurovascular Elements
第四段侧CN(mean ± SD 52.4 ± 2.33 mm vs 46.5 ± 3.71 mm, p < 0.02), CNIX, and CNX(9.91 ± 3.21 mm vs 0.00 ± 0.00 mm, p < 0.01) were exposed in group A than group B. However, shorter portions of CN II (9.31 ± 1.28 mm vs 17.6 ± 6.89 mm, p = 0.009) andV1(26.9±4.62毫米和32.4±1.93毫米,p < 0.03)exposed in group A than group B. There were no statistically significant differences with respect to the other evaluated ipsilateral or contralateral nerves (Fig. 4).

Quantitative comparisons of the CP and PTAP approaches.A:Lengths of the exposed optic nerve (CN II) and tract.B:Lengths of exposed ipsilateral and contralateral CN III.C:Lengths of exposed trochlear, ophthalmic, and maxillary nerves.D:Lengths of exposed cavernous abducens nerve (CN VI), facial nerve (CN VII), vestibulocochlear nerve (CN VIII), and glossopharyngeal and vagus nerves (lower CNs).E:同侧和对侧的长度的暴露posterior cerebral artery (PCA), SCA, and basilar artery (BA).F:Combined areas of the cerebral peduncles and pons exposed lateral (ipsilateral lateral) and medial (ipsilateral medial) to the vertical line that crossed the point where CN III emerges from the interpeduncular fossa, as well as the area of the brainstem past the midline on the contralateral side (contralateral medial).G:Areas of exposed medulla. PMS = pontomedullary sulcus.Error barsindicate SD.
With regard to ipsilateral vessels, longer segments of SCA were exposed in group A than group B (mean ± SD 36.0 ± 4.91 mm vs 25.8 ± 3.55 mm, p < 0.02). Interestingly, group A had less exposure of contralateral SCA than group B (0.00 ± 0.00 mm vs 7.95 ± 3.33 mm, p < 0.01). There were no statistically significant differences between groups with respect to exposure of segments of the basilar artery or the ipsilateral or contralateral posterior cerebral artery.
Brainstem Exposure
没有统计上的显著差异s between groups in terms of the combined areas of the cerebral peduncles and pons that were exposed lateral to the vertical line that crossed the point where CN III emerges from the interpeduncular fossa (p = 0.75;Figs. 3and4). Similarly, the areas of brainstem between that vertical line and the midline, as well as the areas past the midline on the contralateral side, were not different between groups (p = 0.47 and 0.60, respectively).
Although limited exposure of medulla was achieved in group A (mean ± SD 48.1 ± 19.9 mm2), the medulla was not exposed in group B (0.00 ± 0.00 mm2, p < 0.01;Fig. 4).
Angle of Attack
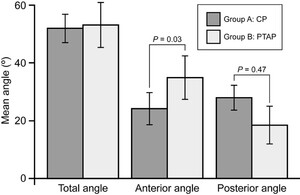
Group A had a smaller anterior angle of attack than group B (mean ± SD 24.1° ± 5.62° vs 34.8° ± 7.51°, p < 0.03) and a larger posterior angle of attack (27.9° ± 4.26° vs 18.3° ± 6.51°, p = 0.047;Fig. 5). There was no difference between groups with respect to the total angle confined by the anterior and posterior rays (51.9° ± 4.90° vs 53.2° ± 7.82°, p = 0.47).

Angles of attack for the CP and PTAP approaches. The PTAP approach allows for a larger angle of attack anterior to the target (intersection of CN VI with Gruber’s ligament), whereas the CP approach allows for a larger angle of attack posterior to the target.Error barsindicate SD.
Discussion
Direct surgical access into and maneuverability within the petroclival region are impeded by the inherent stereo-hindrance created by the osteological and neurovascular structures. Two surgical approaches, CP and PTAP, have been safely utilized to resect PCMs in this location.6,8–10,13We present the first study to morphometrically compare both approaches on an anatomical basis.
Technical Comparison of Surgical Approaches to the Petroclival Region
Two components are common to both approaches: 1) anterior petrosectomy, and 2) trans–Meckel’s cave transtentorial exposure. In both approaches, interdural dissection is used to expose the gasserian ganglion and the lateral wall of the cavernous sinus to variable extents. Similarly, to maximize petroclival exposure, the inferior aspect of V3 is dissected off the underlying petrous ICA and trigeminal impression and reflected as anterior as possible to facilitate removal of the medial petrous apex. In both approaches, theinternal acoustic meatuscan be exposed from porous to fundus, if needed.
Despite these similarities, several key technical maneuvers differentiate the approaches. First, to gain adequate caudal exposure of the petroclival region from the anterolateral perspective afforded by the PTAP approach, extradural anterior clinoidectomy (with or without paraclinoid ICA mobilization) with full mobilization of CN III and drilling of the PCP are often necessary. Such extensive anterior exposure is not required for the CP approach. Second, the CP approach necessities extensive mastoidectomy with mobilization of the sigmoid sinus down to the jugular bulb to adequately expose the sinodural angle and presigmoid dura without violating the semicircular canals. Finally, to adequately mobilize the sigmoid sinus posteriorly with the CP approach, the retrosigmoid and occipital dura above the transverse sinus must be exposed first.
Quantitative Comparison of the CP and PTAP Approaches
To compare exposure between the groups, several neurovascular elements were used as surrogate targets. Our results indicated that the CP approach provides greater exposure of ipsilateral CN IV, CN IX, and CN X, as well as the ipsilateral SCA and medulla. Greater exposure of CN IV with the CP approach could only be achieved by dividing the SPS and resecting the posterior aspect of the tentorium to expose the quadrigeminal cistern. Similarly, exposure of the lower CNs and medulla in a retrolabyrinthine fashion was possible only after disconnecting the transverse-sigmoid junction from the SPS and tentorium and retracting the sinuses posteriorly. Regardless of the extent of drilling into the clivus, the PTAP approach failed to adequately expose CN IX, CN X, and medulla (Figs. 4and6). These findings are consistent with the intraoperative findings of Liao et al.4

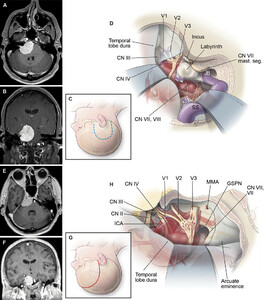
MR images and schematic illustrations of the CP and PTAP approaches for PCM resection.A and B:Axial (A) and coronal (B) MR images with contrast illustrating a PCM that was best managed with the CP approach.C:Schematic illustration of the skin incision used to perform the CP approach.D:Schematic illustration of the surgical view of a PCM that was best approached with the CP approach.E and F:Axial (E) and coronal (F) MR images with contrast illustrating a PCM that was best managed with the PTAP approach.G:Schematic illustration of the skin incision used to perform the PTAP approach.H:Schematic illustration of the surgical view of a PCM that was best approached with the PTAP approach. mast. seg. = mastoid segment. SeeFig. 1for definitions of abbreviations. Used with permission from Barrow Neurological Institute, Phoenix, Arizona (panels C, D, G, and H).
Despite the improved exposure of the medulla and lower CNs obtained with the presigmoid component of the CP approach, it is evident that preservation of the labyrinth hinders the medial extent of the approach. Moreover, any attempt to reach further medially and anteriorly along the brainstem represents, by definition, a transgression of the plane of origin of the lower CNs, which may be at risk during dissection (Fig. 6).
Although both the CP and PTAP approaches provided low enough exposure of the basilar artery to reach the origin of the ipsilateral SCA in each specimen, the mean length of exposed SCA was 28% greater with the CP approach. This improved exposure is directly related to the increased length of the pontomesencephalic segment of the SCA that is visible only from a subtemporal view after posterior division of the tentorium with the CP approach. Interestingly, the PTAP approach allowed for significantly greater exposure of the contralateral SCA (p < 0.01). It was visually evident that the convex shape of the pons, because it opposes the clivus at the level of the SCA origin, obstructed any attempt to reach the contralateral SCA with a posterior approach (such as the CP approach), regardless of the area of drilled clivus. This stereo-hindrance due to the pons limited the length of exposed contralateral SCA to a mean ± SD of 7.95 ± 3.33 mm, but did not completely obstruct exposure.
It was particularly interesting that no statistically significant difference (or even a trend) was found between groups with regard to the combined areas of ipsilateral cerebral peduncles and pons that were exposed lateral (p = 0.75) or medial (p = 0.47) to the vertical line that crossed the point where CN III emerges from the interpeduncular fossa. Similarly, neither approach provided an advantage over the other in terms of contralateral brainstem exposure. These findings are consistent with our clinical observations.
With regard to maneuverability, there was no overall difference between the approaches as measured according to the overall angle of attack (p = 0.47). However, the PTAP approach had better maneuverability than the CP approach in the region anterior to the entrance of CN VI to Dorello’s canal (p = 0.03). Conversely, the posterior angle of attack favored the CP approach (p = 0.05).
Approach Selection
VIDEO 2.A 3D model showing tumors that were best approached with the CP approach or the PTAP approach. Used with permission from Barrow Neurological Institute, Phoenix, Arizona. Clickhereto view.
1) Degree and Direction of Tumor Extension
PCM arises at the tip of the petrous apex medial to the internal auditory canal.2Ichimura and colleagues classified these tumors into four subtypes according to their origin, suspected location of dural attachment, and direction oftrigeminal nervedisplacement.3The subtypes are 1) upper clival, where the tumor attaches medial to CN V and displaces it laterally without cavernous sinus invasion; 2) cavernous, where the tumor originates from the posterior cavernous sinus (medial to CN V) with bilateral extension into theposterior fossaand lateral displacement of CN V; 3) tentorial, where the tumor originates from the tentorium with significant attachment to the petroclival fissure and inferomedial displacement of CN V; and 4) petrous apex, where the tumor attaches to the petrous apex and lies lateral to CN V and medial to the internal auditory canal with superomedial CN V displacement.
In a series of 91 patients, the overall prevalence of tumor invasion into Meckel’s cave was 70.3%, with the lowest prevalence in those with the petrous apex subtype (2 of 8 patients). Hence, anterior transpetrosal and trans–Meckel’s cave exposure is usually an essential component of any surgical approach to large PCMs. The question then becomes whether this step should be combined with a posterior petrosal or an anterolateral transcavernous approach. Our results suggest that, in general, the medial and lateral extents of the tumor in relation to the pons and cerebral peduncles may not be a major determining factor for choosing one approach over the other, because there was no significant difference between groups A and B with respect to the exposed areas of the pons and peduncles (Fig. 4). This finding is consistent with our clinical observations and those of others.4,8However, tumor extension along the rostral-caudal axis is what seems to be important. Specifically, lesions that extend to the lower clivus, compress the medulla, and displace the lower CNs are best managed with the CP approach (Fig. 6). According to our anatomical data, the PTAP approach does not adequately expose this region. However, the CP approach is not a panacea. Reaching further medial and anterior to the medulla through the presigmoid window is difficult because the surgeon would need to operate across the lower CNs, thereby risking iatrogenic injury (Fig. 6). Finally, our results show that the PTAP approach allows for greater exposure of the region anterior to the chiasm.
2) Venous Anatomy
Every patient with PCM must undergo preoperative cerebral venous imaging because such imaging aids in surgical approach planning. A prominent sigmoid sinus and high jugular bulb limit presigmoid exposure with the CP approach. Similarly, if a major draining vein, particularly one on the left side, terminates early into a tentorial sinus instead of entering the region of the transverse-sigmoid sinus junction, the surgeon would have to retract anterior to that vein and thereby limit exposure behind it. In these situations, the PTAP approach may be favored when feasible.
3) Cavernous Sinus Involvement
PCMs often involve the cavernous sinus, as described above. These tumors may infiltrate the ICA21and CNs,22which may explain the high functional cost and recurrence rate associated with aggressive cavernous sinus exploration.23–26Although we do not advocate aggressive exenteration of meningioma from within the sinus proper, like others25,26我们相信,减压的侧壁the cavernous sinus by debulking the tumor in the interdural plane between the meningeal and periosteal dural layers of the lateral wall is beneficial. Hence, in these cases, the PTAP approach may be favored over the CP approach (provided that the tumor does not extend caudally toward the lower CNs as discussed above). Similarly, in patients who require decompression of the optic nerve and canal, anterior clinoidectomy combined with the PTAP approach provides significant value.
4) Hearing Status
It is important to objectively evaluate hearing status bilaterally in each patient with PCM. For patients with functional hearing on only the ipsilateral side of the lesion, performing the CP retrolabyrinthine approach on the functional side may put the only ear with functional hearing at risk of injury. Hence, the PTAP approach would be favored. For patients with significantly compromised hearing on the side of the lesion, the translabyrinthine variation of the CP approach provides greater exposure than the retrolabyrinthine variation without additional risk of morbidity.
Although the abovementioned factors are crucial for surgical approach selection, they cannot be used to predict the final outcome of each operation. As stated by Spetzler’s group in the early 2000s, “Regardless of the surgical approach used, the major determinants of the ability to achieve an excellent resection with a low rate of morbidity are the presence of an arachnoid plane around the tumor, the consistency of the tumor, and the degree of its involvement with critical neurovascular structures.”11Nonetheless, selection of the appropriate surgical approach helps the surgeon to capitalize on these inherent tumor characteristics for the benefit of the patient during surgery.
Finally, regardless of the specific approach used, optimization of postoperative care is key to minimizing morbidities. Provision of adequate hydration after manipulation of the major venous sinuses, as performed with the CP approach, is necessary to prevent thrombosis. Sufficient eye patching and lubrication are mandatory for patients with transient or permanent facial weakness or paralysis. Prism lenses and extraocular muscle surgery may be used to treat patients with permanent cranial nerve palsy.
Literature Review
Considering the excellent yet different exposures afforded with the CP and PTAP approaches, we briefly reviewed the literature and focused on major clinical series that used either approach.
Cho and Al-Mefty wrote one of the first reports to describe the surgical techniques and outcomes of using the CP approach to manage PCM.8In this series, they treated 7 patients, with a 71% rate of gross-total resection and no deaths. One of 6 patients lost hearing in the 1st year after surgery. Preoperatively, 6 patients had House-Brackmann grade I dysfunction and 1 patient had grade II–III dysfunction. At last follow-up, 5 patients had House-Brackmann grade I, 1 patient had House-Brackmann grade II, and 1 patient had House-Brackmann grade V dysfunction. Three patients developed transient abducens palsy, and 1 developed permanent abducens palsy.
In a subsequent series,2764 patients with PCM were evaluated. In this series, the posterior petrosal, CP, anterior petrosal, total petrosal, and transmastoid retrosigmoid approaches were used to treat 27, 15, 11, 4, and 7 patients, respectively. Gross-total resection was achieved in 64% of patients. One postoperative death was related to pulmonary embolism. The mean preoperative Karnofsky Performance Status score was 85.31 (median 90), with a 47% rate of improvement after tumor resection. Preoperatively, 89% of patients had cranial nerve deficit. Of the 54 patients with more than 2 months of follow-up, 39% had persistent deficit. Of note, those with CN V and CNVIIIdeficits were more likely to improve from preoperative status, whereas those with CN VI deficit had a higher risk of sustaining new postoperative injury.
In another series reported by Kusumi et al.,2823个病人接受了CP PCM复位的方法ction, and the rate of gross- or near-total resection was 78.3% and there were no deaths. Eighteen patients had a Karnofsky Performance Status score > 90. Ten patients developed postoperative CN palsy (2 with CN V palsy, 2 with CN VI, 4 with CN VII, and 2 with lower CNs IX–XII). In a study by Mathiesen et al.,1229 patients underwent the CP approach as a part of multimodal therapy for PCM. Simpson grade I and II resections were achieved in 12 patients, grade III in 2 patients, and grade IV in 15 patients. Four patients had preoperative CN III palsy, and 7 had postoperative palsy. All except 1 of these 7 patients with preoperative palsy recovered completely. With regard to CN VI, 3 patients had preoperative palsy, and a total of 6 patients had postoperative palsy. All deficits recovered during the follow-up period. Good facial function (House-Brackmann grades I and II) was obtained in 23 of 29 patients. Useful hearing (Gardner-Robertson hearing scale grades I and II) was preserved in 17 of 23 patients (74%) who underwent hearing preservation surgery. There were no deaths in this series.
To our knowledge, less surgical data are available for the PTAP approach than the CP approach. Liao et al.4reported 18 patients who underwent the PTAP approach for resection of large PCMs. Gross-total resection was achieved in 7 patients (38.9%), near-total resection (> 95%) in 7 patients (38.9%), and subtotal resection (> 85%) in 4 patients (22.2%) (3 with > 90% resection and 1 with > 85%). Although no deaths were reported, all patients developed transient postoperative CN III palsy but recovered within 3 months. Transient abducens palsy was observed in 2 patients and permanent CN IV palsy in 3 patients. The same authors recently published29the findings of their series of 109 patients with skull base meningiomas, of whom 28 had PCMs occupying what they referred to as zone IV (the anterior, middle, and posterior cranial fossa) and were treated with the PTAP approach. Simpson grade I, II, III, and IV resections were achieved in 3.6%, 32%, 17.9%, and 46.4% of patients, respectively. Seven patients developed new cranial nerve deficits and 7 developed strokes. Although the postoperative mortality rate of the entire cohort of 109 patients was 3.7%, the mortality rate of patients with tumor in zone IV was not mentioned.
Future Directions
To our knowledge, this study provides the first direct anatomical comparison of the CP and PTAP approaches. The anatomical advantages and limitations of both approaches were addressed quantitatively. Although the results of this study may guide the approach selection process, the true value of the key elements we identified can only be determined with prospective clinical studies. Future efforts are encouraged to compare the clinical outcomes of both approaches in a prospective manner. Furthermore, future studies—both cadaveric and clinical—should aim to identify strategies to overcome the limitations of each approach.
Conclusions
The CP and PTAP approaches have been used to treat large PCMs. Although each approach is associated with potential complications, true indications for choosing one over the other have not been established. This is the first study to provide objective insight into the advantages and limitations of both approaches from an anatomical perspective. Understanding the technical nuances and anatomical basis of each approach is incumbent on all skull base surgeons and is crucial for selecting the optimal management strategy.
Acknowledgments
We thank the staff of Neuroscience Publications at Barrow Neurological Institute for assistance with manuscript and video preparation.
Disclosures
Dr. Youssef is a consultant for Stryker and receives royalties from Mizuho.
Author Contributions
Conception and design: Labib, Zhao. Acquisition of data: Labib, Zhao, Houlihan, Abramov. Analysis and interpretation of data: Labib, Zhao, Houlihan. Drafting the article: Labib. Critically revising the article: Catapano, Naeem. Reviewed submitted version of manuscript: Lawton, Preul, Youssef. Administrative/technical/material support: Preul. Study supervision: Lawton, Preul, Youssef.
Supplemental Information
Videos
Video 1.https://vimeo.com/500406088.
Video 2.https://vimeo.com/500405708.
References
-
1 ↑
KawaseT,ToyaS,ShiobaraR,MineT.Transpetrosal approach for aneurysms of the lower basilar artery.J Neurosurg.1985;63(6):857–861.
-
2 ↑
Al-MeftyO,FoxJL,SmithRR.Petrosal approach for petroclival meningiomas.开云体育app官方网站下载入口.1988;22(3):510–517.
-
3 ↑
IchimuraS,KawaseT,OnozukaS,et al.Four subtypes of petroclival meningiomas: differences in symptoms and operative findings using the anterior transpetrosal approach.Acta Neurochir (Wien).2008;150(7):637–645.
-
4 ↑
LiaoCH,WangJT,LinCF,et al.Pretemporal trans-Meckel’s cave transtentorial approach for large petroclival meningiomas.Neurosurg Focus.2018;44(4):E10.
-
5 ↑
HakubaA,NishimuraS,JangBJ.A combined retroauricular and preauricular transpetrosal-transtentorial approach to clivus meningiomas.Surg Neurol.1988;30(2):108–116.
-
6 ↑
Al-MeftyO,SmithRR.Combined approaches in the management of brain lesions.In:ApuzzoMLJ, ed.Brain Surgery: Complication Avoidance and Management. Vol2300.Churchill Livingstone;1993.
-
7
Al-MeftyO,AyoubiS,SmithR.The petrosal approach: indications, technique, and results.Acta Neurochir Suppl (Wien).1991;53:166–170.
-
8 ↑
ChoCW,Al-MeftyO.Combined petrosal approach to petroclival meningiomas.开云体育app官方网站下载入口.2002;51(3):708–718.
-
9 ↑
FukushimaT,DayJD,HiraharaK.Extradural total petrous apex resection with trigeminal translocation for improved exposure of the posterior cavernous sinus and petroclival region.Skull Base Surg.1996;6(2):95–103.
-
10 ↑
MillerCG,vanLoveren HR,KellerJT,et al.Transpetrosal approach: surgical anatomy and technique.开云体育app官方网站下载入口.1993;33(3):461–469.
-
11 ↑
BambakidisNC,KakarlaUK,KimLJ,et al.Evolution of surgical approaches in the treatment of petroclival meningiomas: a retrospective review.开云体育app官方网站下载入口.2007;61(5)(suppl 2):202–211.
-
12 ↑
MathiesenT,GerlichA,KihlströmL,et al.Effects of using combined transpetrosal surgical approaches to treat petroclival meningiomas.开云体育app官方网站下载入口.2007;60(6):982–992.
-
13 ↑
DolencVV.A combined epi- and subdural direct approach to carotid-ophthalmic artery aneurysms.J Neurosurg.1985;62(5):667–672.
-
14 ↑
KawaseT,ShiobaraR,ToyaS.Anterior transpetrosal-transtentorial approach for sphenopetroclival meningiomas: surgical method and results in 10 patients.开云体育app官方网站下载入口.1991;28(6):869–876.
-
15 ↑
TripathiM,DeoRC,SuriA,et al.Quantitative analysis of the Kawase versus the modified Dolenc-Kawase approach for middle cranial fossa lesions with variable anteroposterior extension.J Neurosurg.2015;123(1):14–22.
-
16 ↑
TroudeL Jr,CarissimiM Jr,LavieilleJP,RochePH.How I do it: the combined petrosectomy.Acta Neurochir (Wien).2016;158(4):711–715.
-
17 ↑
HanakitaS,WatanabeK,ChampagnePO,FroelichS.How I do it: combined petrosectomy.Acta Neurochir (Wien).2019;161(11):2343–2347.
-
18 ↑
LabibMA,BorbaMoreira L,ZhaoX,et al.The side door and front door to the upper retroclival region: a comparative analysis of the open pretemporal and the endoscopic endonasal transcavernous approaches.J Neurosurg.2020;133(6):1892–1904.
-
19 ↑
SekerA,InoueK,OsawaS,et al.Comparison of endoscopic transnasal and transoral approaches to the craniovertebral junction.World Neurosurg.2010;74(6):583–602.
-
20 ↑
FunakiT,MatsushimaT,Peris-CeldaM,et al.Focal transnasal approach to the upper, middle, and lower clivus.开云体育app官方网站下载入口.2013;73(2 Suppl Operative):ons155–ons191.
-
21 ↑
KotapkaMJ,KaliaKK,马丁内斯AJ,SekharLN.Infiltration of the carotid artery by cavernous sinus meningioma.J Neurosurg.1994;81(2):252–255.
-
22 ↑
LarsonJJ,vanLoveren HR,BalkoMG,努力工作JM Jr.Evidence of meningioma infiltration into cranial nerves: clinical implications for cavernous sinus meningiomas.J Neurosurg.1995;83(4):596–599.
-
23
DeMonteF,SmithHK,al-MeftyO.Outcome of aggressive removal of cavernous sinus meningiomas.J Neurosurg.1994;81(2):245–251.
-
24
SindouM,WydhE,JouanneauE,et al.Long-term follow-up of meningiomas of the cavernous sinus after surgical treatment alone.J Neurosurg.2007;107(5):937–944.
-
25 ↑
GozalYM,AlzhraniG,Abou-Al-ShaarH,et al.Outcomes of decompressive surgery for cavernous sinus meningiomas: long-term follow-up in 50 patients.J Neurosurg.2020;132(2):380–387.
-
26 ↑
KlingerDR,FloresBC,LewisJJ,BarnettSL.The treatment of cavernous sinus meningiomas: evolution of a modern approach.Neurosurg Focus.2013;35(6):E8.
-
27 ↑
AlmeftyR,DunnIF,PravdenkovaS,et al.True petroclival meningiomas: results of surgical management.J Neurosurg.2014;120(1):40–51.
-
28 ↑
KusumiM,FukushimaT,梅塔AI,et al.Tentorial detachment technique in the combined petrosal approach for petroclival meningiomas.J Neurosurg.2012;116(3):566–573.
-
29 ↑
ChenSC,LinCF,LiaoCH,et al.The pretemporal trans-cavernous trans-Meckel’s trans-tentorial trans-petrosal approach: a combo skill in treating skull base meningiomas.J Neurooncol.2020;146(3):407–416.