Thepurpose of this paper is to provide an answer to the controversial question of the opening of the central canal (CC) into thefourth ventricle. About a century ago, J. T. Wilson, professor of anatomy at the University of Sidney, stated: “when one turns to the standard text-books in search of the precise manner in which the CC of the medulla oblongata actually expands into the cavity of the fourth ventricle, only vague and general statements are, for the most part, met with.”1,2
Nowadays, some vagueness still endures about the visual appearance of the opening of the CC into the fourth ventricle. Despite countless pathological studies illustrating the shape, size, and variations of the whole CC along the spinal cord and medulla, a definite anatomical picture of the opening of the CC in the fourth ventricle, or "apertura canalis centralis" (ACC), as elegantly named by Theodor Ziehen, as far as we know is still unavailable.1,2
This minute anatomical structure of the size of tenths of a millimeter is deeply concealed within the median fissure and the obex. Therefore, the ACC is understandably masked while examining cadaveric specimens, even with microsurgical techniques. Actually, meticulous and exhaustive microsurgical studies cite the location of the ACC, but they do not show its morphology as it would be perceived to the direct sight of the viewer.3,4
Even during the microsurgical exploration of the inferior triangle of the fourth ventricle, the dot-like orifice of the ACC remains undetectable, being hidden by the obex.5–7Besides, in this view the axis of the CC is directed caudocranially, whereas the ideal point of observation for the ACC, as suggested by Wilson and others, should be from the top.1,2,8,9Such a strategic viewpoint, achieved with difficulty during microsurgical operations in theposterior fossa, could be easily afforded through a transaqueductal approach toward the fourth ventricle using flexible scopes.10,11Remarkably, this technique does not alter the physiological conditions of CSF ventricular pressure, which probably are the critical factor in keeping the ACC open.
We reviewed our endoscopic experience of transaqueductal fourth ventricle exploration to define the frequency of visualization of the ACC, its morphological and morphometric details, and its precise position in the calamus, as well as its relationship with thearea postrema. These data, beyond their anatomical value, could be useful in microneurosurgical approaches, adding another useful landmark to the puzzle of structures in the inferior triangle of the floor of the fourth ventricle.
开云体育世界杯赔率
In this study, the analyses of the morphology and morphometry of the ACC in the fourth ventricle were based on a revision of recorded cases in which an endoscopic transaqueductal fourth ventricle inspection was performed. From May 1994 until December 2019, the authors performed 834 endoscopic ventricular procedures with 194 transaqueductal fourth ventricle explorations. All patients gave informed consent, which was approved by the institutional review board. In this group of patients, we reviewed the digitally recorded cases, and we selected 44 cases fitting the purpose of this research (Supplemental Fig. 1).The mean age of the patients was 63.2 years (range 19–79 years), although a pediatric patient aged 3 years was also included (Table 1).
Patient demographics and ACC anatomical features
| Pt No. | Pt Sex/Age (yrs) | Disease | ACC Shape | ACC Size (µm) | ACC-Obex (mm) | Ratio | AP Size | ACC Position* | Tools |
|---|---|---|---|---|---|---|---|---|---|
| Group A | |||||||||
| 1 | F/71 | IVH | Lanceolate | 700 × 200 | 0.5 | 1 | 3.0 × 0.5 | C++ | FL |
| 2 | M/61 | IVH | Lanceolate | 700 × 250 | 1.5 | 1.5 | 2.5 × 0.5 | C+ | |
| 3 | M/76 | IVH | Lanceolate | 750 × 250 | 3 | 2 | 1 × 0.75 | C+ | |
| 4 | M/54 | IVH | Lanceolate | 500 × 200 | 2.5 | 2.5 | 2 × 0.5 | C++ | |
| 5 | M/78 | IVH | Round double | 400 | 2.5 | 4 | 3 × 0.5 | IC | FL |
| 6 | M/63 | IVH | Lanceolate | 700 × 200 | 3 | 3 | 2 × 1 | C+ | FL |
| 7 | M/76 | IVH | Ovoid lanceolate | 700 × 400 | 3 | 4 | 2.5 × 1 | C+++ | PICA |
| 8 | F/67 | IVH | Ovoid lanceolate | NA | NA | 1 | NA | NA | |
| 9 | M/63 | IVH | Triangular | 400 | 3 | 4.5 | 2.0 × 1 | C+ | FL |
| 10 | M/66 | IVH | Lanceolate | 800 × 200 | 1.5 | 1.5 | 2 × 0.5 | C+++ | FL |
| 11 | M/79 | IVH | Lanceolate | 500 × 150 | 1 | 2 | 3 × 0.75 | C+ | |
| 12 | F/55 | IVH | Lanceolate | 500 × 150 | 1.5 | 3 | NA | NA | |
| 13 | M/63 | IVH | Ovoid lanceolate | NA | NA | 2 | NA | NA | |
| 14 | F/69 | IVH | Lanceolate | NA | NA | 2.5 | NA | NA | |
| 15 | M/72 | IVH | Lanceolate | 500 × 100 | 1.5 | 3 | 2 × 1 | C+ | |
| 16 | M/56 | IVH | Lanceolate | 750 × 150 | 1.5 | 2 | 3 × 1 | C+ | FL |
| Group B | |||||||||
| 17 | F/65 | MOCA | Round | 150 | 2 | 2 | NA | NA | |
| 18 | F/54 | MOCA | Triangular | 500 × 200 | 1 | 2 | NA | NA | |
| 19 | F/26 | MOCA | Lanceolate | 700 × 150 | 1.5 | 2 | 2.0 × 0.5 | C+ | PICA |
| 20 | M/65 | MOCA | Quadrangular | 300 × 500 | 1 | 3 | 2.5 × 0.3 | IC | FL |
| Group C | |||||||||
| 21 | M/60 | FVOO | Lanceolate | 750 × 250 | 2 | 3 | 3 × 0.5 | C+ | |
| 22 | M/63 | FVOO | Lanceolate | 500 × 150 | 1.5 | 3 | 1 × 1 | C+ | |
| 23 | F/19 | FVOO | Lanceolate | 1000 × 500 | 4 | 4 | 2 × 0.5 | C+++ | |
| 24 | M/58 | FVOO | Lanceolate | 500 × 150 | 2.5 | 3 | 2 × 2 | C+ | |
| 25 | F/65 | FVOO | Round | 400 | 2 | 5 | NA | NA | COAG |
| 26 | M/66 | FVOO | Round | 500 | 1.5 | 3 | 2 × 0.5 | C+++ | FL |
| 27 | F/79 | FVOO | Roundish | 400 | 2 | 5 | 2 × 0.5 | C++ | FL |
| 28 | M/3 | FVOO | Round | 150 | 1.5 | 8 | 2 × 1 | C++ | COAG |
| 29 | F/47 | FVOO | Lanceolate | 500 × 200 | 4 | 6 | 1.5 × 1.5 | C+++ | PICA |
| 30 | F/65 | AC | Round double | 400 | 2 | 2 | 2.5 × 0.5 | C++ | |
| Group D | |||||||||
| 31 | F/60 | NPH | Triangular | 300 | 0.5 | 3 | 2.5 × 0.3 | C+ | |
| 32 | M/76 | NPH | Roundish triangle | 300 | 1.5 | 5 | 2 × 0.5 | C+ | |
| 33 | F/66 | NPH | Quadrangular | 300 × 400 | 0.5 | 1 | 1.5 × 0.3 | IC | |
| 34 | M/71 | NPH | Triangular | 300 × 150 | 0.5 | 2 | 2 × 0.3 | C+ | |
| 35 | M/78 | NPH | Roundish | 200 | 1.5 | 3 | 3 × 0.3 | IC | PICA |
| 36 | M/71 | NPH | Triangular | 300 | 1 | 3 | 1.5 × 1.2 | C+++ | PICA, FL |
| 37 | M/63 | NPH | Lanceolate | 500 × 200 | 1 | 2 | 2 × 0.3 | C+ | |
| 38 | M/72 | NPH | Round | 300 | 1.5 | 3 | 1 × 0.3 | C+ | FL |
| 39 | M/73 | NPH | Round | 150 | 1 | 5 | NA | NA | |
| 40 | M/64 | NPH | Round | 300 | 1.5 | 4 | 1 × 1.5 | C+ | PICA, FL |
| 41 | M/72 | NPH | Round | 250 | 1.5 | 8 | 0.5 × 2.5 | C+ | PICA |
| 42 | F/74 | NPH | Round | 250 | 0.5 | 2 | 0.5 × 1.5 | C+ | |
| 43 | M/74 | NPH | Round | 300 | 1 | 3 | NA | NA | |
| 44 | F/67 | NPH | Rhomboid | 500 × 250 | 0.5 | 1 | 2 × 0.5 | C++ | PICA |
AC = arachnoid cyst of the cisterna magna; ACC = apertura canalis centralis; ACC-obex = distance between ACC and obex; AP = area postrema; C = cranial; COAG = monopolar coagulator; FL = fluorescein sodium; FVOO = fourth ventricular outlet obstruction; IC = intracommissural; IVH = intraventricular hemorrhage; MOCA = membranous obstruction of the cerebral aqueduct; NA = not available; NPH = normal pressure hydrocephalus; PICA = posterior inferior cerebellar artery; pt = patient; ratio = ratio between the ACC-obex distance and the longitudinal segment of the ACC.
Values were not obtained from metric units, but from the geometrical extension of the considered segments. Consequently, the resulting absolute number can be considered less biased.
ACC position is assessed with reference to the AP commissure. C indicates “cranial” in relation to the AP commissure, and C+, C++, and C+++ indicate progressive distance between the ACC and AP commissure.
16例(A组)与tetr承认aventricular hemorrhage and underwent endoscopic blood clot aspiration, and 4 patients (group B) underwent aqueductoplasty because of hydrocephalus due to a membranous occlusion of the cerebral aqueduct, followed by inspection of the fourth ventricle. These groups could reasonably have included patients with normal fourth ventricle anatomy. In fact, we expected little structural variation in the patients with intraventricular hemorrhage, which was endoscopically aspirated at an early stage (within 24 hours). On the other hand, patients who underwent aqueductoplasty were expected to have a normal fourth ventricle by definition. Ten patients presented with hydrocephalus due to blocked fourth ventricle (group C), and 14 patients were affected by normal pressure hydrocephalus (NPH; group D). Of course, in the group C patients the fourth ventricle anatomy was considered likely to express pathological variation. Although not routinely performed, endoscopic exploration of the fourth ventricle in cases of probable or possible NPH is helpful to rule out any undiagnosed obstruction of the fourth ventricular outlets. Actually, some of our patients initially diagnosed with NPH and for whom MRI did not conclusively exclude obstruction proved to be affected by fourth ventricular outlet obstruction (FVOO).13
Transaqueductal Approach to the Fourth Ventricle
技术采用经验的描述loration of the fourth ventricle with flexible scopes has been provided in previous papers, in which we focused on the endoscopic anatomy of the cerebral aqueduct and the fourth ventricle, and magendieplasty procedures.10,12–14Through a right precoronal burr hole positioned 2 cm anterior to thecoronal sutureand 1.5–2 cm from the midline, a 14-Fr peel-away catheter is used to cannulate thelateral ventricle. The flexible endoscope (2.5- or 3.9-mm diameter; Karl Storz) is then introduced into thethird ventricleand deflexed posteriorly toward the adytum of the aqueduct (Fig. 1).Trespassing the aqueduct with an adequately sized flexible scope is safe, with the compliance of the aqueduct being surprisingly broad and allowing complete navigation of the fourth ventricle as well as some surgical actions when deemed necessary.

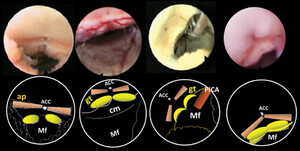
A:Endoscopic trajectory to the calamus scriptorius.B:Inferior triangle of the fourth ventricle from a posterior point of view. The calamus scriptorius is hidden by the obex.C:Inferior triangle of the fourth ventricle from a postero-superior point of view, and after removing the membrane of the obex. ac = ala cinerea; ACC = apertura canalis centralis; ap = area postrema; as = acoustic striae; at = acoustic tubercle; cp = cerebellar peduncles; ct = cuneate tubercle; fs = funiculus separans; gt = gracile tubercle; ht = hypoglossal triangle; ob = obex; vt = vagal triangle. Theasteriskindicates the intraventricular portion of the gracile tubercle. Copyright Alberto Feletti. Published with permission. Figure is available in color online only.
Morphometry and Anatomical Items
Measurements were taken by the first author and were double-checked by a colleague. The main morphological and morphometrical items included prevalence, shape, color, and size of the ACC; its relationship with the area postrema; and its position compared to the obex (Fig. 2).

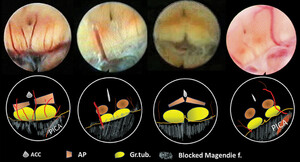
Paradigmatic morphology of the calamus scriptorius.A:Endoscopic view.B:Schematic illustration. Thedashed blue lineshows the caudal border of the fourth ventricle coinciding with the Magendie foramen. Gracile tubercles (gt) are partially intraventricular and partially extraventricular. The funiculus separans (fs) is the only structure crossing the median line in this area. Thearrowheadsindicate the apertura canalis centralis. ap = area postrema; cm = cisterna magna; ms = median sulcus; ob = obex. Figure is available in color online only.
To better define the prevalence of the ACC, we classified our observations as no evidence (A0); indirect evidence, which was defined as a significant depression without signs of any dimple (A1); and clear evidence (A2).
The shape and color of the ACC were recorded for descriptive purposes. The ACC shape was defined as round, rhomboid, triangular, or lanceolate along themedian sulcus. The color was described as dark and inhomogeneous dark, depending on the apparent patency and CSF circulation.
Due to the retrospective nature of the study, the size of the ACC could be only indirectly assessed through a comparison with known instruments and structures. Photos showing the tip of an endoscopic monopolar coagulator (1 mm) or the extremity of a Fogarty balloon (1 mm) used in 2 cases of magendieplasty turned out to be useful for providing an acceptable estimate of the millimetric measurements of the area of the calamus (Fig. 3).A second group of comparative elements included some readily recognizable anatomical structures, the sizes of which have been well assessed in the anatomical literature. In 8 cases the loop of the公元后下rebellar artery(PICA) was visible, and according to Kawashima et al., a mean value of 1.6 mm was assigned to the diameter of this artery.15Furthermore, in most cases the contour of the area postrema was visible either due to its tenuous orange color starting in the midline close to the ACC or after enhancement following intravenous fluorescein sodium injection (12 cases). The value of 2.5 mm was given to the length and 0.5 mm to the depth of the area postrema.11In a consistent number of cases, the gracile tubercles or clavae were clearly visible. Their approximate size was about 2.5 mm.16The inner congruence of these reference values, when evaluated together in each patient, improved the plausibility of our computations. In all cases, comparative measurements were made while taking care to position the tip of the endoscope consistently at about the same distance from the obex, in order to minimize the error related to differences in relative magnification.

比较使用单极coagulat形态测量学or (upper panel) and other known anatomical structures (lower panel).Ch. pl = choroid plexus; Gr. tub. = gracile tubercles; PICA = posterior inferior cerebellar artery. Figure is available in color online only.
The relations of the ACC with the area postrema, the obex, and the Magendie foramen were classified as “caudal,” “at the same level” (i.e., “intracommissural”), and “rostral.”
Results
Prevalence
The ACC was clearly observed in all of the procedures of fourth ventricle inspection. The perception of the ACC was classified as A1 in only 2 cases, with the remaining being classified as A2. Two cases presented a double ACC, one close to the commissure of the area postrema and the second along the median sulcus 0.5–1 mm more cranially.
Shape
VIDEO 1.编译的内窥镜爱的探索mus scriptorius. The following situations are shown: 1) lanceolate ACC in a patient with membranous obstruction of the cerebral aqueduct; 2) ovoid lanceolate ACC in a patient with IVH; 3) lanceolate ACC in a patient with IVH, in whom fluorescein sodium was used; 4) round ACC in a patient with FVOO; 5) rhomboid ACC in a patient with NPH; and 6) endoscopic exploration during a microsurgical posterior fossa approach in a patient in the prone position; in this case, the ACC is not clearly seen, probably because the hydrostatic CSF pressure is lacking. Copyright Alberto Feletti. Published with permission. Clickhereto view.
The same shape was also observed in some patients with obstructive hydrocephalus (group C). The round shape was common in the NPH group (group D) (Figs. 4–6,Video 1).The latter profile is associated with the disappearance of the sulcus medianus, along with the flattening of the medial eminences, which in turn leads to a rounded ACC in the calamus. These progressions could be easily framed within the atrophic processes peculiar to the slowly evolving NPH. In the group of blocked fourth ventricle egresses, the calamus scriptorius is somehow overstretched, and consequently, all of the neural structures are elongated. Similarly, the ACC is also distorted, often taking the form of a narrow, uniformly dark triangle.

Some examples of ACC in group A and B cases (likely to include patients with normal fourth ventricle anatomy), with the predominant lanceolate shape.Asterisksindicate the extraventricular part of the gracile tubercles.Arrowheadsindicate the apertura canalis centralis. gt = gracile tubercle; Mf = Magendie foramen. Figure is available in color online only.

The shape of the ACC in NPH (group D). The oval shape is predominant, with atrophic features depending on chronic process. ACC = apertura canalis centralis; ap = area postrema; cm = cisterna magna; gt = gracile tubercle; Mf = Magendie foramen; PICA = posterior inferior cerebellar artery. Figure is available in color online only.

In obstructive hydrocephalus (group C), the stretching caused by intraventricular pressure spreads apart the structures of the calamus scriptorius, elongating them. This process gives the ACC a prevalent lanceolate shape. ACC = apertura canalis centralis; AP = area postrema; f. = foramen; Gr.tub. = gracile tubercles; PICA = posterior inferior cerebellar artery. Figure is available in color online only.
Color
The color of the caudal part of the ACC was always dark, indicating that the CC emerges from the depth of the calamus scriptorius into the fourth ventricle. The dark color looked attenuated along the median sulcus.
Size
The average size value of the ACC in groups A and B (with possible normal anatomy) was 600 × 250 µm (range 1000–200 × 700–150 µm). The sizes in group C (obstructive hydrocephalus) and group D (NPH) were 500 × 250 µm (range 150–1000 × 150–400 µm) and 250 × 200 µm (range 500–230 × 300–150 µm), respectively.
Relation With the Obex
The distances between the caudal margin of the ACC and the obex were about 1.8 mm (3–0.9 mm) in groups A and B, 2.1 mm (range 4–0.5 mm) in group C, and 1 mm (range 1.5–0.5 mm) in group D.
Relation With the Area Postrema
区域postrema本身显示重大variations in length and width. It had an oval silhouette in only 2 cases. In the remaining cases, the main variation was in the width of its two strips at the level of the ACC, or just 100 µm caudal to it. The commissure joining the two wings of the AP constitutes the caudal edge of the ACC in almost all normal and pathological cases. In 4 cases, the AP seemed to pass through the commissure, penetrating it. The commissure of the AP showed enhancement after sodium fluorescein injection only in 5 of 12 cases, indicating that it is a different tissue (Fig. 7,Video 1).This tissue was identified as the funiculus separans, a strip of tissue mentioned by anatomists as cranially delimiting the area postrema and separating it from the ala cinerea, although we could not identify the latter in any of our cases.

The region of the area postrema as it appears with white light (upper panels) and fluorescence mode (lower panels).The size and shape of the area postrema are quite variable. Its relationship with the ACC (arrowheads) is consistent, given that the ACC is always more cranial as it is contoured by the funiculus separans. Figure is available in color online only.
Patient demographic and ACC anatomical features are summarized inTable 1.
Discussion
The CC is a thin CSF duct, 50–100 µm in size, which runs along the spinal cord and the medulla oblongata, ending up in the calamus scriptorius of the fourth ventricle.17,18Similarly to the contiguous ventricular cavities filled with CSF, the CC is lined by ependymal cells sharing the common embryonal origin of the primitive neural tube.
Although the discovery of the CC is credited to Estienne in the 16th century,19only histological studies dating back to the 19th century, along with further investigations by Gardner20and Milhorat,21have raised the question of the pathophysiology of this microscopic channel, as well as of its controversial relation to the development of syringomyelia. At birth, the CC is pervious and remains patent in 100% of infants age 14 months or younger,17and afterward, it goes through progressive alterations and even regression.22Kasantikul et al. showed that the CC was closed in all adults with normal ventricular size, but also in 94% of persons with various degrees of hydrocephalus.23
More recent studies have confirmed that the CC undergoes focal obstructions, strictures, and extended obliterations due to the hyperproliferation of the ependymal layer.24,25Results of current investigations suggest that such hyperproliferation could be related to the activation of the ependymal layer, strictly connected to subependymal stem cell niches, and in turn involved in repairing spinal cord damages.26As is now known, this feature is common to the entire subventricular system of adult mammalians with the same biological characteristics of all stem cells, including the ability for self-renewal and differentiation into neurons and astrocytes or oligodendrocytes.27It is supposed that within these segmental CC obstructions the CSF circulates freely, with these luminal proliferations being only occasional and temporary.25The CC runs through two totally distinct structures of the nervous system: the medulla oblongata and the spinal cord. In a study including 232 cases, Milhorat et al. showed that CC strictures or obliterations have a very different incidence in medulla and equivalent spinal cord sections, occurring more than twice in the latter.25This finding suggests that, differently from the spinal cord, CSF circulates more freely in the medullary CC. In this perspective, the medullary CC could also be regarded as the aqueduct (or ventricle) of the medulla oblongata rather than the simple extension of the CC of the spinal cord. Caudally, the CC extends 5–6 cm beyond the conus medullaris into the filum terminale. At the level of the conus medullaris, the CC sometimes enlarges and becomes visible as the terminal ventricle of Krause (fifth ventricle).28
The ACC: A Forbidden Fruit for Human Eyes?
Theapertura canalis centralisis the specific name in the old-fashioned literature for the opening of the CC. It represents the upper end of the CC emerging into the fourth ventricle. The results based on our endoscopic experience show that during the exploration of the fourth ventricle the ACC can always be clearly seen as the apparently pervious opening of the CC, as suggested by the dimness of the tiny dimple opening in the calamus scriptorius. The achievement of anatomical pictures and the tentative morphometry of the ACC along with its closer neural structures represent the motivation and may be the main value of our investigation.
As previously mentioned, compared with the large numbers of images of many detailed histological studies, almost nothing is known about the appearance of the ACC to the human eye.4,15Although laboratory studies have demonstrated great accuracy, images of the calamus scriptorius are poorly visible,1,2and the same limitations can be noticed in subsequent research.8Even the scanning electron micrograph of the caudal floor of the fourth ventricle shows a quite different scenario compared to that given by endoscopy.9
In operative neurosurgical reports, the ACC is rarely cited. In 1962, Lindstrom and Brizzee coagulated the areae postremae in 5 patients with intractable vomiting.29These authors certainly had to thoroughly inspect the calamus, but they do not cite the ACC, which is positioned just a bit caudal to the wings of the area postrema. Gardner treated syringomyelia closing up the ACC by using small pieces of muscle or cotton wisps on it.20In a figure of his paper representing the operative scenario of an open posterior fossa in the sitting position, he shows a nerve hook hanging from the obex. He comments that by so doing without a microscope, he could intercept the entrance of the “funnel shaped” CC, whose pictures were not provided anyway. We selected these important historical contributions since they clearly demonstrate that visibility of the ACC is really problematic. Even now, the ACC sounds somehow mythical to neurosurgeons, ascertained by anatomists but barely seen in surgical practice. Some intrinsic factors could impede the direct perception of the ACC: its submillimetric size, its position in the median sulcus very close to the obex, and its cranial orientation. Moreover, adequate intraventricular pressure may be the sine qua non condition to maintain the perviousness of the channel. During the inspection of fresh anatomical specimens, the AAC could simply collapse due to the absence of CSF and its pressure, which keeps the canal open. In such situations, the soft walls of the ACC flatten in the groove of the median sulcus, vanishing and merging in the white floor of the medullary surface.
The same flattening of the ACC could happen during microsurgical suboccipital telovelar approaches, where the detection of the ACC is prevented by the obex and the caudocranial perspective of the surgeon’s observation, but also by the almost complete CSF leakage decreasing the intracranial pressure and causing a collapse of the tiny groove of the ACC.
The craniocaudal viewpoint and the preservation of CSF dynamics are critical for the success of the endoscopic transaqueductal approach in obtaining and recording direct images of the ACC. Moreover, once the aqueduct is passed, flexible endoscopes have the advantage of proceeding at ground level along the floor of the fourth ventricle with a craniocaudal direction and a perpendicular/orthogonal point of observation toward the foramen of Magendie, exactly in front of the calamus scriptorius and the ACC (Fig. 1).Finally, it is noteworthy that this privilege of observing the ACC is not due to the endoscopic procedure per se, given that endoscopic caudocranial trans–Magendie foramen approaches have failed to provide images of the ACC.5,30,31
Morphology and Morphometry of the ACC
The opening of the CC in the fourth ventricle is possibly due to the divergence of the gracile fascicles under which the CC runs, being limited only by the obex. From a deep central position in the spinal cord and the medulla, the CC progressively emerges at the apex of the inferior triangle of the fourth ventricle along the median sulcus.1,2,8,9This peculiar geometrical configuration could also explain the lanceolate shape of the ACC often observed in the study of normal cases.
Once the gracile fascicles in the region of the calamus diverge from the midline in a V-shaped manner, they end with a swelling called the gracile tubercle or clava due to the subjacent nucleus gracilis and constituting the true caudal border of the fourth ventricle, upon which the band of the obex is tended (Fig. 1).The two clavae,通常由obex连体,杜曾被观察到ring our endoscopic observation as two white areas delimiting the calamus scriptorius with an average size of 2.5 mm and a notch in between. The CC, becoming free of the overlying mechanical constraints of the gracile fascicles, progressively reaches the dorsal surface, emerging from the center of the medulla into the ventricle, cranially to both the obex and the tissue connecting the areae postremae.8Therefore, proceeding cranially from the band of the obex, it is possible to locate the gracile tubercles within no more than a couple of millimeters, along with the connecting tissue of the two areae postremae sharply bordering the ACC caudally.
The thin region connecting the areae postremae plays an important role in contouring the ACC. Wislocki and Putnam, exactly a century ago, defined the area postrema as “two mounds of extremely vascular tissue converging caudally until they unite forming as they do the roof of the CC.”32In our opinion, this is a good definition of the position of the ACC. Moreover, in that way the area postrema is strategically located at the opening of the CC, similarly to other circumventricular organs that are positioned at the border of contiguous CSF chambers.8Nonetheless, there are some uncertainties about the nature of the tissue caudally contouring the ACC (indicated as tissue connecting the areae postremae, or intercinereal commissure as Wilson called it1,2).在我们的经验中,荧光素注入enhanced the tissue connecting the areae postremae in 5 of 12 cases (Fig. 7).Therefore, this tissue seems not to belong to the area postrema; alternatively, it could coincide with the funiculus separans, a subtle thread delimiting the area postrema itself cranially and separating it from the ala cinerea (Fig. 2).Nonetheless, we could not endoscopically identify the entire anatomical pathway, as others have done in laboratory specimens.4,8,9
Milhorat and Miller passionately remarked that “the obex is not synonymous with the upper end of the central canal,”33since in a sagittal section the caudal border of the ACC coincides with the connecting tissue of the areae postremae and not with the obex, positioned more caudally and then having nothing to do with the ACC. Our contribution confirms these statements, possibly identifying the submillimetric area between the obex and the area postrema–ACC complex with a small portion of the gracile tubercles or clavae. Actually, as clearly shown by direct endoscopic inspections, the surface of the gracile tubercles is partially extraventricular and partially intraventricular, covered by ependyma (Fig. 2).The area postrema is certainly positioned caudally to the ACC, but it is not the hindmost structure of the ventricular floor, as the Latinpostremus, superlative of the adjectiveposterus, could indicate.34The term is imprecise since between the area postrema and the band of the obex there are two small crescent-shaped areas (lunulae) of nervous tissue, sized approximately 1.8 mm (range 1.0–2.5 mm), corresponding in our opinion to the intraventricular part of the gracile tubercles, reaching the considerable extension of 4 mm in hydrocephalic cases. Their rounded shape, the indentation in between, and the stretch itself observed in blocked fourth ventricles seem to confirm this suggestion. In normal cases, the gracile fascicles and tubercles, soon after their diversion, can fill up just an exiguous fraction of the space between the area postrema and obex, but when ventricular pressure increases, the obex as the rearmost wall of the ventricle becomes squeezed back, unveiling the tubercles underneath or clavae.
The size of the ACC is also of great interest. It seems to far exceed that of the CC of the spinal cord. In the normal subset group, the more frequent lanceolate or triangular shape average size is 250 µm (range 150–700 µm) for the transverse diameter in the more caudal part and 600 µm (range 700–1000 µm) for the length along the median sulcus. Therefore, its dimension is in the range of visibility to the naked eye. In the context of the elongate form of the ACC, a more caudal part can be distinguished, corresponding to the bulk of the emerging CC, and a more cranial extension along a sort of groove progressively carved in the median sulcus, possibly since the fetal stage of life. This opening of the CC extending along the median sulcus creates the wide ranges of morphological variations we observed in the group with normal anatomy (Fig. 4).In a schematic way, there is a constant caudal component corresponding to the opening of the CC. Its shape is generically slightly rounded, squared, or rhomboid. This part is always caudally sharply limited by the commissure of tissue connecting the areae postremae, a condition quite comparable to that of the aqueduct bordered dorsally by the posterior commissure. The cranial component of the ACC with the elongated form developing cranially is subject to certain variability, resembling a triangle or having a lanceolate shape. In 2 cases, a short membrane partially roofing the channel along the median sulcus determined a double opening of the ACC, one caudal and the other some 100 µm cranially.
Pathological processes also induce variations in shape. In NPH cases, the median sulcus becomes flattened and the cranial elongation of the ACC disappears. The lanceolate form changes and shifts to various triangular profiles and even to a nude well-rounded shape, similarly to the pathological aspect of the aqueduct in the same disease (Fig. 5).These variations should be better studied and interpreted in relation to intraventricular pressure or to atrophic processes or impaired CSF dynamics in hydrocephalus. On the other hand, in the case of obstructive hydrocephalus the intraventricular pressure causes a stretch on the entire calamus, which reshapes the ACC into a triangular contour that otherwise could be rounded (Fig. 6).
Limitations of the Study
This study has some limitations that must be taken into account. First, this is a retrospective study and there might be a selection bias due to the fact that we had to exclude those cases that had only a partial exploration of the fourth ventricle, or for which only an unclear or corrupted video recording was available. Second, the measurement techniques are obviously not optimal, as they are based on comparison with other anatomical structures and surgical tools. Despite our attempt to minimize the error related to differences in relative magnification, the measurements have a margin of inaccuracy as there are no ways to obtain a direct measure of anatomical structures with an endoscope inside the fourth ventricle.
Conclusions
Endoscopy, as microsurgery, has contributed to our ability to observe the innermost anatomical structures, providing details otherwise difficult to achieve, such as the triangular recess, the circumventricular organs, the posterior part of the roof of the third ventricle, and so on.
The opening of the CC in the fourth ventricle or ACC is a consistent structure of the calamus scriptorius. The literature takes for granted the presence at the end of the ventricle of the ACC, albeit to our knowledge only flexible endoscopy, with the evidence of the images we have shown, could reveal the ACC perhaps for the first time, answering questions posed by Wilson a century ago.1,2With these premises, further microsurgical studies could refine and complete the topography of the calamus, providing more reliable landmarks for use by neurosurgeons.
Disclosures
The authors report no conflict of interest concerning the materials or methods used in this study or the findings specified in this paper.
Author Contributions
Conception and design: Longatti. Acquisition of data: Feletti, Longatti, Fiorindi. Analysis and interpretation of data: Feletti, Longatti, Marton, Sala. Drafting the article: Feletti, Longatti. Critically revising the article: all authors. Reviewed submitted version of manuscript: all authors. Approved the final version of the manuscript on behalf of all authors: Feletti. Study supervision: Longatti.
Supplemental Information
Videos
Video 1.https://vimeo.com/502297176.
Online-Only Content
Supplemental material is available with the online version of the article.
Supplemental Fig. 1.//www.prize-show.com/doi/suppl/10.3171/2020.12.JNS203649.
References
-
1 ↑
WilsonJT.On the anatomy of the calamus region in the human bulb; with an account of a hitherto undescribed “Nucleus postremus.”: Part I.J Anat Physiol.1906;40(pt 3):210–241.
-
2 ↑
WilsonJT.On the anatomy on the calamus region in the human bulb; with an account of a hitherto undescribed “Nucleus postremus.”: Part II.J Anat Physiol.1906;40(pt 4):357–386.
-
3 ↑
MussiAC,RhotonALJr.Telovelar approach to the fourth ventricle: microsurgical anatomy.J Neurosurg.2000;92(5):812–823.
-
6
TanrioverN,UlmAJ,RhotonALJr,YasudaA.Comparison of the transvermian and telovelar approaches to the fourth ventricle.J Neurosurg.2004;101(3):484–498.
-
7
TomaselloF,ContiA,AngileriFF,CardaliS.Telo-velar approach to fourth-ventricle tumours: how I do it.Acta Neurochir (Wien).2015;157(4):607–610.
-
8 ↑
DuvernoyH,KoritkéJG,MonnierG,JacquetG.Vascularization of the area postrema and of the dorsal side of the medulla oblongata in the human. Article in French.Z Anat Entwicklungsgesch.1972;138(1):41–66.
-
9 ↑
LeslieRA.Comparative aspects of the area postrema: fine-structural considerations help to determine its function.Cell Mol Neurobiol.1986;6(2):95–120.
-
10 ↑
LongattiP,FiorindiA,FelettiA,et al.Endoscopic anatomy of the fourth ventricle.J Neurosurg.2008;109(3):530–535.
-
11 ↑
LongattiP,PorzionatoA,BasaldellaL,et al.The human area postrema: clear-cut silhouette and variations shown in vivo.J Neurosurg.2015;122(5):989–995.
-
12
LongattiP,FiorindiA,FelettiA,BarattoV.Endoscopic opening of the foramen of Magendie using transaqueductal navigation for membrane obstruction of the fourth ventricle outlets. Technical note.J Neurosurg.2006;105(6):924–927.
-
13 ↑
LongattiP,FiorindiA,MartinuzziA,FelettiA.Primary obstruction of the fourth ventricle outlets: neuroendoscopic approach and anatomic description.开云体育app官方网站下载入口.2009;65(6):1078–1086.
-
14
LongattiP,FiorindiA,PerinA,MartinuzziA.Endoscopic anatomy of the cerebral aqueduct.开云体育app官方网站下载入口.2007;61(3)(suppl):1–6.
-
15 ↑
KawashimaM,RhotonALJr,TanrioverN,et al.Microsurgical anatomy of cerebral revascularization. Part II: posterior circulation.J Neurosurg.2005;102(1):132–147.
-
16 ↑
CiołkowskiM,SharifiM,TarkaS,CiszekB.Median aperture of the fourth ventricle revisited.Folia Morphol (Warsz).2011;70(2):84–90.
-
17 ↑
Petit-LacourMC,LasjauniasP,IffeneckerC,et al.Visibility of the central canal on MRI.Neuroradiology.2000;42(10):756–761.
-
18 ↑
SakerE,HenryBM,TomaszewskiKA,et al.The human central canal of the spinal cord: a comprehensive review of its anatomy, embryology, molecular development, variants, and pathology.Cureus.2016;8(12):e927.
-
19 ↑
MarkatosK,ChytasD,KorresD,et al.Charles Estienne (1504-1564): his life, work, and contribution to anatomy and the first description of the canal in the spinal cord.World Neurosurg.2017;100:186–189.
-
20 ↑
GardnerWJ.Hydrodynamic mechanism of syringomyelia: its relationship to myelocele.J Neurol Neurosurg Psychiatry.1965;28:247–259.
-
21 ↑
MilhoratTH,CapocelliALJr,AnzilAP,et al.Pathological basis of spinal cord cavitation in syringomyelia: analysis of 105 autopsy cases.J Neurosurg.1995;82(5):802–812.
-
22 ↑
NetskyMG.Syringomyelia; a clinicopathologic study.AMA Arch Neurol Psychiatry.1953;70(6):741–777.
-
23 ↑
KasantikulV,NetskyMG,JamesAEJr.Relation of age and cerebral ventricle size to central canal in man. Morphological analysis.J Neurosurg.1979;51(1):85–93.
-
24 ↑
GöritzC,FrisénJ.Neural stem cells and neurogenesis in the adult.Cell Stem Cell.2012;10(6):657–659.
-
25 ↑
MilhoratTH,KotzenRM,AnzilAP.Stenosis of central canal of spinal cord in man: incidence and pathological findings in 232 autopsy cases.J Neurosurg.1994;80(4):716–722.
-
26 ↑
WeissS,DunneC,HewsonJ,et al.Multipotent CNS stem cells are present in the adult mammalian spinal cord and ventricular neuroaxis.J Neurosci.1996;16(23):7599–7609.
-
27 ↑
PanayiotouE,MalasS.Adult spinal cord ependymal layer: a promising pool of quiescent stem cells to treat spinal cord injury.Front Physiol.2013;4:340.
-
29 ↑
LindstromPA,BrizzeeKR.Relief of intractable vomiting from surgical lesions in the area postrema.J Neurosurg.1962;19:228–236.
-
30 ↑
Di IevaA,KomatsuM,KomatsuF,TschabitscherM.Endoscopic telovelar approach to the fourth ventricle: anatomic study.Neurosurg Rev.2012;35(3):341–349.
-
31 ↑
MatulaC,ReinprechtA,RoesslerK,et al.Endoscopic exploration of the IVth ventricle.Minim Invasive Neurosurg.1996;39(3):86–92.
-
32 ↑
WislockiGB,PutnamTJ.Further observations on the anatomy and physiology of the areae postremae.Anat Rec.1924;27(3):151–156.
-
33 ↑
MilhoratTH,MillerJI.The obex is not synonymous with the upper end of the central canal. Letter.Pediatr Neurosurg.1994;21(1):112.
-
34 ↑
SarikciogluL,YildirimFB.Area postrema: one of the terms described by Magnus Gustaf Retzius.J Hist Neurosci.2008;17(1):109–110.