Duringthe last 2 decades, neuroendoscopy has become an important adjunct in the management of intraventricular brain tumors.8我t is especially useful for lesions that obstruct the CSF pathways because it allows one, in a minimally invasive way, to biopsy the tumor and control hydrocephalus. The reliability and effectiveness of endoscopic biopsies have been established by several studies.7,9The help of neuronavigation can also allow safe and effective biopsies in cases of normally sized ventricles.9On the contrary, experience with endoscopic tumor removal is limited. Usually only cystic and small tumors are debulked or totally resected through purely endoscopic approaches.1–3,5–7,10,14,15Most experience has been gained with colloid cysts,4,6but other tumors are also suitable for endoscopic removal, such as subependymal giant cell astrocytomas (SEGAs), other exophytic low-grade gliomas, central neurocytomas, small choroid plexus tumors, and purely intraventricular craniopharyngiomas.1,3,5,11,16
切除的病变是妥协的lack of instruments suitable for removing such lesions as well as problems with visibility because of bleeding. Therefore, to be considered for endoscopic removal, tumors should present the following characteristics: moderate to low vascularity, soft consistency, smaller than 2 cm in diameter, association with hydrocephalus, low grade histologically, and situated in the lateral or third ventricle.16
Here we report our initial experience with the utilization of an endoscopic ultrasonic aspirator in 12 pediatric patients affected by intraventricular tumors. To the best of our knowledge after a search of the literature, intraventricular tumor resection using an ultrasonic aspirator via an endoscopic channel has been reported only once, in an adult patient.15This tool has the potential to expand the spectrum of intracranial pathologies treatable by modern neuroendoscopic means.
开云体育世界杯赔率
Twelve pediatric patients (10 male, 2 female), ages 1–15 years old, underwent surgery via a purely endoscopic approach using a Gaab rigid endoscope (Karl Storz GmbH) and an endoscopic ultrasonic aspirator (Sonoca, Söring GmbH). The technical features of the endoscopic ultrasonic aspirator are fully described elswhere.13The basic hardware and setup are shown inFig. 1. The characteristics of the patients are summarized inTables 1–3. The patients were divided into 3 groups according to tumor type: purely intraventricular tumors, intraparaventricular tumors, and suprasellar tumors.

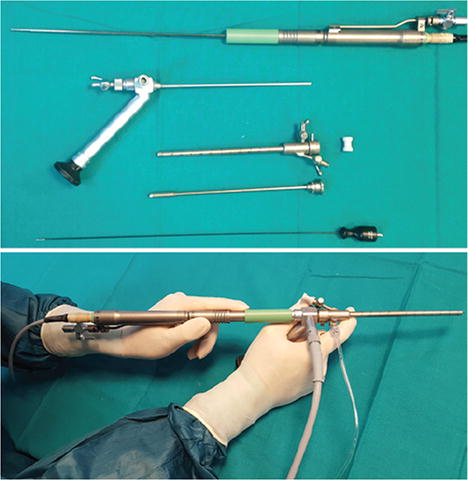
Upper:Elements necessary for the use of an endoscopic ultrasonic aspirator (from the top): handpiece of the ultrasonic Sonoca aspirator, Gaab rigid endoscope, cannula for ventricular entry, trocar of the cannula, and bipolar coagulator.Lower:Setup of the system and the preferred way of handling: the right hand holds the endoscope (high-definition camera attached below; not shown), the left hand holds the aspirator handpiece, and the left index finger controls the depth of introduction of the handpiece through the working channel. The light cable is attached on the right, as is an infusion set for continuous irrigation with warm Ringer's lactate solution through the working channel. The aspiration circuit is attached at the posterior connector of the handpiece (not shown). Figure is available in color online only.
Purely intraventricular tumors
| Case No. | Sex, Age (mos) | Histological Diagnosis | Lesion Location | No. of Procedures Performed | Bur Hole | Hydrocephalus | Extent of Removal | Residual Disease/FU | Op Time (mins) |
|---|---|---|---|---|---|---|---|---|---|
| 1 | F, 67 & 68 | MB | 1) rt frontal horn, 2) lt frontal horn | 2 | 1) rt coronal, 2) lt coronal | VP shunt 6 mos later | Complete (100%) | Dead due to progressive disease 6 mos after 2nd procedure | 40 & 44 |
| 2 | M, 16 | ATRT | 3rd ventricle | 1 | Rt coronal | Previous ETV | Complete (100%) | Dead due to progressive disease 6 mos after endoscopic procedure | 37 |
| 3 | M, 89 | SEGA | Lt foramen of Monro | 1 | Lt coronal | Monoventricular (lt lat, treated w/tumor removal) | Near complete (>95%) | Stable residue at 24-mo FU | 76 |
| 4 | M, 179 | SEGA | Rt foramen of Monro | 1 | Rt coronal | None | Near complete (>95%) | Stable residue at 18-mo FU | 120 |
| 5 | M, 12 | Pineal anlage tumor | Pineal, 3rd ventricle, lat ventricles | 1 | Lt frontal, hairline | ETV | Complete (100%) | 指化疗后完全删除w /普洛古莱sive metastatic lesion | 60 |
| 6 | M, 74 | 我mmature teratoma | Pineal, 3rd ventricle | 1 | Rt frontal, hairline | ETV (failed), VP shunt | Partial (<10%) | Tumor completely removed via transtentorial approach 1 mo after endoscopic procedure | 80 |
chemo = chemotherapy; ETV = endoscopic third ventriculostomy; FU = follow-up; MB = medulloblastoma; VP = ventriculoperitoneal.
我ntraparaventricular tumors
| Case No. | Sex, Age (mos) | Histological Diagnosis | Lesion Location | No. of Procedures Performed | Bur Hole | Hydrocephalus | Extent of Removal | Residual Disease/FU | Op Time (mins) |
|---|---|---|---|---|---|---|---|---|---|
| 7 | M, 90 | Low-grade glioneuronal tumor | Trigone of rt lat ventricle | 2 | 1) rt occipital, 2) rt occipital | No | 1) partial (60%), 2) subtotal (>95%) | Stable tumor residue at 6-mo FU | 1) 60, 2) 65 |
| 8 | M, 135 | Low-grade glioneuronal tumor | Rt thalamus | 1 | Rt frontal | No | Enlarged biopsy (20%) | Subdural hygroma 1 wk after op; microsurgical removal of thalamic tumor 3 mos after endoscopic procedure | 50 |
Suprasellar/third ventricular tumors
| Case No. | Sex, Age (mos) | Histological Diagnosis | Lesion Location | No. of Procedures Performed | Bur Hole | Hydrocephalus | Extent of Removal | Residual Disease/FU | Op Time (mins) |
|---|---|---|---|---|---|---|---|---|---|
| 9 | M, 132 | Craniopharyngioma (at presentation) | Suprasellar/3rd ventricle | 2 | Rt coronal | No | Near total (>90%) | Progression of residue, microsurgical removal 6 mos after endoscopic procedure | 65 |
| 10 | M, 48 | Residue of craniopharyngioma | 3rd ventricle | 1 | Rt coronal | No | Complete (100%) | No recurrence at 12-mo FU (patient underwent radiosurgery) | 30 |
| 11 | F, 36 | Pilocytic astrocytoma | Suprasellar/3rd ventricle | 1 | Rt coronal | No | Partial (<20%) | Shrinkage of tumor after 1 yr of chemo (SIOP 2004, LGG protocol) | 45 |
| 12 | M, 168 | Pilocytic astrocytoma | Suprasellar/3rd ventricle, cyst invading lt thalamus | 1 | Rt coronal | Yes | Partial (<20%), good marsupialization of cyst | Stable at 5-mo FU | 50 |
LGG = low-grade glioma; SIOP = Società Italiana di Oncologia Pediatrica (Italian Society of Pediatric Oncology).
The first group included 2 patients with intraventricular metastases from high-grade tumors (medulloblastoma and atypical teratoid rhabdoid tumor [ATRT]), 2 with SEGAs, 1 with an immature teratoma of the pineal region/posterior third ventricle, and 1 with a small remnant of a giant pineal anlage tumor that had been removed in 3 earlier microsurgical procedures. In the 2 cases of metastases, a multidisciplinary team including neurosurgeons, oncologists, and radiotherapists established the indication for surgery. The patient with the medulloblastoma (Case 1) presented with small intraventricular nonenhancing lesions in both lateral ventricles (Fig. 2) 1 year following initial treatment and a first operation, consisting of gross-total tumor removal, high-dose chemotherapy, and radiotherapy to the posterior fossa, according to an “infant” protocol. The aim of surgery was to confirm the diagnosis and to remove all visible tumor before salvage therapy including bone marrow suppression. The patient in Case 2 was a 1-year-old boy who presented with 3 small nodules in the posterior part of the third ventricle following total removal of a fourth ventricle/aqueduct ATRT (Fig. 3). Surgery was scheduled to eradicate the tumor before radiotherapy. The patients in Cases 3 and 4 were affected by tuberous sclerosis; the patient in the former case presented with symptomatic monolateral hydrocephalus due to SEGA (Fig. 4), whereas the patient in the latter case presented with an asymptomatic, incidentally discovered intraventricular SEGA that showed signs of enlarging on neuroimaging (Fig. 5). The patient in Case 5 underwent surgery to remove the residue of a large, previously surgically treated pineal anlage tumor. The patient in Case 6 was affected by an immature teratoma of the pineal region.

Case 1. Five-year-old girl affected by medulloblastoma with intraventricular metastases. Axial contrast-enhanced T1-weighted MR images obtained before (left) and after (right) endoscopic removal of a tumoral nodule (arrow) in the left lateral ventricle. Figure is available in color online only.

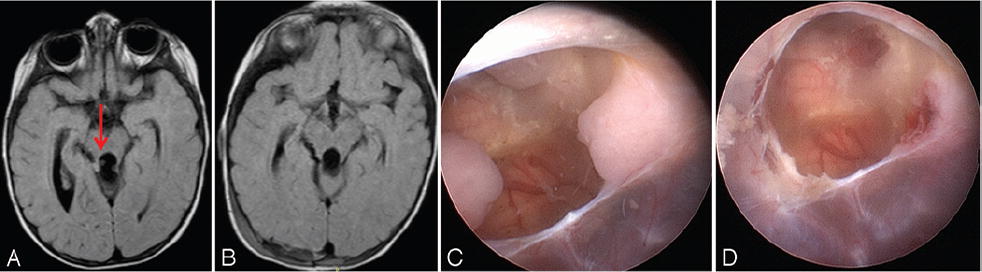
Case 2. One-year-old boy affected by ATRT. Axial FLAIR MR image showing small tumor nodules (arrow) in the posterior part of the third ventricle (A). Postoperative image (B). Endoscopic view of the nodules before (C) and after (D) endoscopic removal. Figure is available in color online only.

Case 3. Seven-year-old boy affected by tuberous sclerosis and monolateral hydrocephalus due to intraventricular SEGA. Contrast-enhanced T1-weighted MR images obtained before (left) and after (right) endoscopic removal of a left foramen of Monro tumor and septostomy.

Case 4. Fifteen-year-old boy affected by tuberous sclerosis and bilateral foramen of Monro SEGAs. Contrast-enhanced T1-weighted MR images obtained before (left) and after (right) endoscopic removal of the largest tumor in the right lateral ventricle. The smaller tumor is under observation.
The second tumor group included 2 patients who presented with low-grade intraparaventricular lesions, one in the trigone of the right lateral ventricle (Fig. 6) and the other in the right thalamic region. Both tumors were incidentally discovered following minor head injury. Surgery was scheduled to obtain tissue for diagnosis.

Case 7. Seven-year-old boy with an incidentally discovered intraparaventricular tumor.A:Coronal MR image showing the right ventricle tumor.B:Coronal MR tractography image showing the pyramidal tract that surrounded the tumor.C:Arrowindicates the endoscopic trajectory through an occipital bur hole, with the patient in the lateral position.D:Postoperative image obtained following the first operation, showing complete removal of the intraventricular portion of the tumor.E:Postoperative image obtained after the second operation.F:Stable tumor residue at 6-month follow-up. Figure is available in color online only.
The group with suprasellar tumors included 2 patients with craniopharyngiomas and 2 patients with an optic pathway glioma (OPG). All tumors expanded into the third ventricle. The patient in Case 9 (Figs. 7and8) underwent surgery at presentation, with the aim of removing the cystic portion of the tumor and as much as possible of the solid portion. The patient in Case 10 underwent surgery to remove a small intraventricular residue following open surgery. The patient in Case 11 (Figs. 9and10), who harbored an OPG, underwent surgery to obtain tissue for diagnosis and to remove tumor from the foramina of Monro and the aqueduct to restore the CSF pathways before chemotherapy. The patient in Case 12 had a cystic relapse of an OPG.

Case 9. Eleven-year-old boy with a craniopharyngioma. Midsagittal DRIVE MR images obtained at presentation (left) and postoperatively (right).

Case 9. Intraoperative images showing laser coagulation of the choroid plexus and tumor capsule (A), ultrasonic fragmentation and aspiration of the cystic portion (B), and ultrasonic dissection and aspiration of the solid portion (C). Figure is available in color online only.

Case 11. Three-year-old girl with OPG. Midsagittal DRIVE MR images obtained at presentation (left) and immediately postoperatively (right), the latter showing removal of the upper part of the tumor, with clearing of the foramina of Monro and the aqueduct.

Case 11. Intraoperative images showing visualization of the tumor in the third ventricle and its initial debulking with biopsy forceps (A) as well as ultrasonic fragmentation and aspiration of the tumor (B) until the aqueduct was free of tumor (C). Figure is available in color online only.
Surgical Technique
Five hydrocephalic patients presented with large ventricles. In 7 patients the ventricles were of normal size. In all cases, the endoscopic trajectory and ventricular access were guided by electromagnetic neuronavigation (Stealth-Station AxiEM, Medtronic). We chose an endoscopic trajectory that avoided eloquent structures but allowed the best possible view.
Nine patients underwent surgery via a precoronal bur hole while supine. In 2 cases the bur hole was frontal, at the level of the hairline. One patient underwent surgery via an occipital approach while in the lateral position (Fig. 6C). The endoscopic technique was similar in all cases.
Fragments of tumor were removed with biopsy forceps and collected for pathological evaluation (Fig. 10A). Finally, the tumor was aspirated with the ultrasonic aspirator (Figs. 8B and Cand10B and C). This tool allows both fragmentation and aspiration of tumor tissue; however, aspiration must be carefully adjusted to low levels with intermittent irrigation to avoid ventricle collapse. Twenty percent of aspiration power as well as 20% of cavitation power was used as the standard initial setting. Irrigation with Ringer's lactate solution at 36 C° was started as soon as aspiration was initiated to prevent ventricular collapse and was continued for the entire time that aspiration was on. An occasional increase in cavitation power was necessary, especially for the calcified craniopharyngioma (Video 2) and SEGAs, but every effort was made to keep the aspiration power as low as possible.VIDEO 1.Case 7. Surgical video of 2 neuroendoscopic procedures performed to remove a large intraparaventricular tumor of the right lateral ventricle in a 7-year-old boy. The tumor was partially removed via an occipitoparietal bur hole with the patient in the left lateral position. Use of the endoscopic ultrasonic aspirator is extensively shown. Histological analysis revealed a low-grade glioneuronal tumor. One month later the patient underwent reoperation via the same approach. Subtotal tumor removal was achieved. Copyright Giuseppe Cinalli. Published with permission. Clickhereto view.
如果增加愿望力量是必要的,照顾was taken to use it intermittently in frequent alternation with irrigation to avoid ventricular collapse or excessive decreases in intraventricular pressure. This was particularly necessary in normally sized ventricles, in which 3–4 seconds of aspiration was sufficient to induce complete ventricular collapse and complete loss of visibility. In such cases irrigation could quickly reestablish a ventricular volume sufficient to allow good visibility, safe navigation, and safer surgical maneuvers. In all of the procedures and throughout their duration, the image quality offered by the high-definition camera always allowed excellent magnification of the tumor-brain or tumor-ependyma interface. This allowed safe removal of tumor tissue without neurological complications. The only limiting factors were very hard tumor consistency, resistance to cavitation or aspiration even with very high settings (Case 6), or intraoperative loss of precise neuronavigation, which would necessitate endoscopic reoperation 3 months later (Case 7). Otherwise, even with calcified tumors (SEGAs or craniopharyngiomas), careful adaptation of aspiration and cavitation power allowed for delicate dissection of pathological tissue from ependymal or large vascular structures such as thalamostriate veins during SEGA removal. All removed tissue was captured in a collection chamber and was used for pathological analysis. Usually only minor bleeding occurred and thus was controlled with irrigation alone. Rarely, major vessels were coagulated preferably with a thulium contact laser or, as an alternative in cases of persistent bleeding, with monopolar electrocautery. The procedure was stopped when all intraventricular exophytic portions of the tumor were removed; accidental violations of ependyma or brain parenchyma did not occur (Figs. 8Cand10C).VIDEO 2.Case 9. Surgical video of a neuroendoscopic procedure performed to remove a large solid and cystic craniopharyngioma in an 11-year-old boy presenting with intracranial hypertension, hydrocephalus, papilledema, and vision loss. The tumor cyst is aspirated first with removal of a large part of the cyst wall. Then the solid calcified part is progressively reduced to leave only the component infiltrating the hypothalamus. Copyright Giuseppe Cinalli. Published with permission. Clickhereto view.
Results
Histological evaluation of the collected material—obtained using biopsy forceps and aspirated with an ultrasonic aspirator—was diagnostic in all cases. The duration of the operations ranged from 30 to 120 minutes. There were no major complications; only 1 patient developed subdural hygroma several days after the procedure. All procedures were performed by the senior author. Followup ranged from 2 to 24 months (mean 9.4 months).
我llustrative Cases
Purely Intraventricular Tumors
The patients in Cases 3 (Fig. 4) and 4 (Fig. 5), both affected by SEGA in the foramen of Monro, underwent near-total removal (> 95% of the lesion) of their intraventricular tumors (Table 1). Both lesions were very hard and calcified, but our tool proved to be effective throughout the procedure in the fragmentation and aspiration of tumor tissue. In both cases the foramen of Monro was released by tumor. In 1 case the associated monolateral hydrocephalus resolved. The residual tumor in Case 4 was stable at the 18-month follow-up.
我ntraparaventricular Tumors
我n Case 7 (Video 1) the endoscopic transventricular approach was chosen because the pyramidal tract was considered to be at risk in a transcortical microsurgical approach, as suggested by MR tractography (Fig. 6B). The patient underwent surgery in the lateral position, via an occipital approach (Fig. 6C). The intraventricular portion of the tumor was completely removed at the first operation (Fig. 6D and E),但是没有切除intraparenchymal部分t attempted because of the perceived loss of neuronavigational precision during surgery, probably attributable to the shift induced by removal of the intraventricular mass. Histological analysis revealed a low-grade glioneuronal tumor with papillary aspects. The patient underwent surgery again 3 months later via the same approach and the same endoscopic technique under navigational control to remove the parenchymal portion of the tumor, and subtotal removal was achieved with stable residual tumor at the 6-month follow-up (Fig. 6F).
Suprasellar Tumors
The patient in Case 9 (Video 2) underwent near-total removal of a mixed cystic and solid craniopharyngioma (Fig. 7). The tool proved to be effective even in the fragmentation and aspiration of the calcified component of the tumor (Fig. 8). The small residue on the floor of the third ventricle progressed, and the patient underwent surgery again via a subfrontal microsurgical approach.
Discussion
Purely endoscopic resection of intraventricular tumors is difficult and time consuming. In a recent meta-analysis by Barber et al.,240 studies focusing on endoscopic resection of intraventricular tumors were reported. More than 650 cases were identified. The results of this meta-analysis were encouraging: complete or near-complete resection was achieved in 75.0% of the patients. Almost all cases had been surgically treated using standard endoscopic instrumentation such as forceps, monopolar or bipolar coagulation, and suction catheters. In only a few cases was an assistive device, designed for rapid tumor dissection and removal, used. Even though objective data regarding the overall benefits of these devices are lacking, the surgeons who use the devices describe them as useful.10,12,15
The first experience with ultrasonic aspiration in neuroendoscopy was reported by Oertel et al. in 2008.13The accuracy and reliability of tissue aspiration, issues with visibility, and the application of suction were tested in cadaveric porcine brains. The authors achieved reliable, accurate, and effective aspiration of the paraventricular brain parenchyma under water at the wall of the lateral ventricle. They observed vigorous water movements with great suction force. Therefore, they recommended minimal suction strong enough to suck tissue debris but low enough to avoid obscuring the operative field. They also found a correlation between applied aspiration force and aspiration depth. Especially at low amplitudes, the brain parenchyma was often not completely destroyed and aspirated tissue pieces remained adherent to the brain parenchyma. At higher amplitudes, very effective and accurate tissue aspiration was reported.
These authors13also tested an ultrasonic aspirator in 5 clinical endoscopic applications: 2 cases of pituitary macroadenoma (via an endonasal transsphenoidal approach), 2 cases of third ventricular hemorrhages, and 1 case of cystic craniopharyngioma. In all clinical cases, amplitudes of 60%–80% were applied. Aspiration of soft pituitary adenoma tissue was achieved without difficulty or complications. Moreover, intraventricular hematoma evacuation and cyst wall perforation were performed without any difficulty. In particular, these investigators emphasized that the operative field was not visually obscured. They concluded that the ultrasonic aspirator tip could be useful in expanding the spectrum of endoscopic procedures, but they remained concerned about whether this device could be used to aspirate firm meningiomas or calcified fibrous tumors. Additionally, in pituitary tumor surgery, caution must be exercised with regard to the risk of carotid artery injury, pituitary gland damage, and diaphragm perforation.
Selvanathan et al.15described the first case of solid tumor removed using an ultrasonic aspirator. It was a small tumor inside the aqueduct (low-grade glioneuronal tumor). They reported a temporary loss of visibility during the procedure as a result of air bubbles caused by the ultrasonic aspirator. Formation of these bubbles was caused by a cavitation effect due to rapid motion of the ultrasonic aspirator tip and by the transmission of ultrasonic energy to the surrounding fluid medium. During this period of obstructed visibility, the authors reported that the aqueduct was contused—possibly causing the temporary fourth nerve palsy.
这种现象不会发生在我们的经验。我n normally sized ventricles, however, continuous irrigation was frequently necessary as soon as the endoscope was inserted into the ventricle to attain minimal enlargement of the ventricular system, allowing safer navigation and tumor resection. Increased aspiration caused ventricular collapse. To avoid this, the suction force was set at a minimum and an assistant was always ready to manually clamp the suction tube in case the ventricular walls were going to collapse. Moreover, the aspiration system was kept closed during navigation maneuvers, whereas continuous irrigation with warm Ringer's lactate solution through an endoscopic channel was performed to facilitate debris removal and keep a positive intraventricular pressure and larger ventricular chambers. When an instrument is changed, one should be careful to prevent fluid escape from the ventricles; otherwise, visibility could be obscured. We do not usually remove the ultrasonic aspirator from the working channel of the endoscope; instead, we withdraw it only partially and advance the other instrument (for example, a monopolar coagulator) through a second working channel. A specially designed plastic cap is applied at the entrance of the accessory working channel of the Gaab endoscope to introduce small flexible instruments (laser fiber or coagulation wire) through the channel, avoiding fluid loss. Obviously, as with all new instruments, it is important for the surgeon to become familiar with the settings of the endoscopic ultrasonic aspirator before using it, especially the aspiration settings.
The main limitation of this technique, a limitation that can often prevent complete resection, is the restricted angle of introduction of the ultrasonic aspirator, which is coaxial with the endoscope. The dissection and aspiration of large firm tumors may be impossible, especially when working in small ventricles.
Thus, in our opinion, the main indication for this technique is small tumors in which radical resection can be easily achieved or larger tumors in which radical resection may not be necessary, such as OPG (Figs. 9and10) and some types of intraventricular craniopharyngiomas infiltrating the hypothalamus in which subtotal resection followed by radiotherapy is the treatment of choice.
Conclusions
我n this preliminary pediatric experience, the use of an endoscopic ultrasonic surgical aspirator proved to be safe and effective in completely removing small and midsized intraventricular tumors, even calcified lesions, and in achieving partial tumor resection to obtain a valid histological sample and restore CSF pathways. With realistically applicable safe settings, complete removal of tumors with a hard texture (for example, teratoma) is still impossible. Further experience and technological improvements may increase in the near future the possible indications for this tool in the management of intraventricular tumors.
References
-
1 ↑
AbdullahJ,CaemaertJ:Endoscopic management of craniopharyngiomas: a review of 3 cases.Minim Invasive Neurosurg38:79–84,1995
-
2 ↑
BarberSM,Rangel-CastillaL,BaskinD:Neuroendoscopic resection of intraventricular tumors: a systematic outcomes analysis.Minim Invasive Surg2013:898753,2013
-
3 ↑
CinalliG,SpennatoP,CianciulliE,FiorilloA,Di MaioS,MaggiG:The role of transventricular neuroendoscopy in the management of craniopharyngiomas: three patient reports and review of the literature.J Pediatr Endocrinol Metab19:Suppl 1341–354,2006
-
4 ↑
DecqP,Le GuerinelC,BrugièresP,DjindjianM,SilvaD,KéravelY,et al.:Endoscopic management of colloid cysts.开云体育app官方网站下载入口42:1288–1296,1998
-
5 ↑
DelitalaA,BrunoriA,ChiappettaF:Purely neuroendoscopic transventricular management of cystic craniopharyngiomas.Childs Nerv Syst20:858–862,2004
-
6 ↑
GaabMR,SchroederHWS:Neuroendoscopic approach to intraventricular lesions.J Neurosurg88:496–505,1998
-
7 ↑
GiannettiAV,AlvarengaAY,de LimaTO,PedrosaHA,SouweidaneMM:Neuroendoscopic biopsy of brain lesions: accuracy and complications.J Neurosurg122:34–39,2015
-
9 ↑
LeeMH,KimHR,SeolHJ,ShinHJ:Neuroendoscopic biopsy of pediatric brain tumors with small ventricle.Childs Nerv Syst30:1055–1060,2014
-
10 ↑
LekovicGP,GonzalezLF,Feiz-Erfan我,RekateHL:Endoscopic resection of hypothalamic hamartoma using a novel variable aspiration tissue resector.开云体育app官方网站下载入口58:1 SupplONS166–ONS169,2006
-
11 ↑
MacarthurDC,BuxtonN,VloeberghsM,PuntJ:The effectiveness of neuroendoscopic interventions in children with brain tumours.Childs Nerv Syst17:589–594,2001
-
12 ↑
MohantyA,ThompsonBJ,PattersonJ:最初的经验endoscopic side cutting aspiration system in pure neuroendoscopic excision of large intraventricular tumors.World Neurosurg80:655.e15–655.e21,2013
-
13 ↑
OertelJ,KraussJK,GaabMR:Ultrasonic aspiration in neuroendoscopy: first results with a new tool.J Neurosurg109:908–911,2008
-
14 ↑
OiS,ShibataM,TominagaJ,HondaY,ShinodaM,TakeiF,et al.:Efficacy of neuroendoscopic procedures in minimally invasive preferential management of pineal region tumors: a prospective study.J Neurosurg93:245–253,2000
-
15 ↑
SelvanathanSK,KumarR,GooddenJ,TyagiA,ChumasP:Evolving instrumentation for endoscopic tumour removal of CNS tumours.Acta Neurochir (Wien)155:135–138,2013
Disclosures
The authors report no conflict of interest concerning the materials or methods used in this study or the findings specified in this paper.
Author Contributions
Conception and design: Cinalli. Acquisition of data: Imperato, Mirone, Di Martino, Nicosia. Analysis and interpretation of data: Cinalli, Aliberti. Drafting the article: Spennato. Critically revising the article: Cinalli. Reviewed submitted version of manuscript: Cinalli. Approved the final version of the manuscript on behalf of all authors: Cinalli. Administrative/technical/material support: Mirone, Ruggiero. Study supervision: Cinalli. Performed all surgeries described as first surgeon: Cinalli.
Supplemental Information
Videos
Video 1.https://vimeo.com/188997280.
Video 2.https://vimeo.com/188997555.